Table of Contents
Advertisement
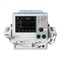
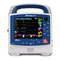
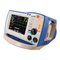
Chapter 1 General Information
The use of accessories, transducers, and cables other than those specified in this manual may
•
result in increased emissions or decreased immunity of the ZOLL M2 monitor/defibrillator.
To ensure protection against the effects of defibrillator discharge, use only ZOLL-approved
•
accessories.
Always perform a functional test of internal paddles prior to use.
•
Always inspect the unit for damage if it has been dropped.
•
Only authorized personnel should use the Supervisor menus.
•
If uncertain about the accuracy of any measurement, first check the patient's vital signs by
•
alternate means, and then make sure the monitor is functioning correctly.
Do not use the ZOLL M2 in the presence of oxygen-rich atmospheres, flammable
•
anesthetics, or other flammable agents (such as gasoline). Using the unit in such
environments might cause an explosion.
While the device can operate normally in an environment up to 50° C, continued monitoring
•
or treatment should be performed in a more normal temperature environment to reduce the
chance of heat-related injury to the patient.
Before disposing of equipment, in order to avoid contaminating or infecting personnel, the
•
environment, or other equipment, it is important to disinfect and decontaminate the monitor/
defibrillator and any accessories and to remove the batteries. Dispose of the device and
accessories in accordance with your country's regulations for equipment containing
electronic parts.
When other devices are used with the ZOLL M2 unit, their potential equalization terminals
•
can be connected together to eliminate potential differences between different devices.
Do not perform preventative maintenance service on the ZOLL M2 unit while it is
•
connected to a patient.
Do not modify the ZOLL M2 unit.
•
Portable RF communications equipment (including peripherals such as antenna cables and
•
external antennas) should be used no closer than 30 cm (12 inches) to any part of the
ZOLL M2 unit, including cables specified by the manufacturer. Otherwise, degradation of
the performance of this equipment could result.
ECG Monitoring
Implanted pacemakers can cause the heart rate meter to count the pacemaker rate during
•
incidents of cardiac arrest or other arrhythmias. Dedicated pacemaker detection circuitry
may not detect all implanted pacemaker spikes. Check the patient's pulse; do not rely solely
on heart rate meters. Patient history and physical examination are important factors in
determining the presence of an implanted pacemaker. Pacemaker patients should be
carefully observed. See "Pacemaker Pulse Rejection:" on page A-4 of this manual for
disclosure of the pacemaker pulse rejection capability of this instrument.
Use only ECG electrodes that meet the AAMI standard for electrode performance
•
(AAMI EC-12). Use of electrodes not meeting this AAMI standard could cause the ECG
trace recovery after defibrillation to be significantly delayed.
Prior to attempting synchronized cardioversion, ensure the ECG signal quality is good and
•
that sync markers are displayed above each QRS complex.
Do not place electrodes directly over an implanted pacemaker.
•
The ZOLL M2 unit detects ECG electrical signals only. It does not detect a pulse (effective
•
circulatory perfusion). Always verify pulse and heart rate by physical assessment of the
patient. Never assume that the display of a nonzero heart rate means that the patient has a
pulse.
1-14
www.zoll.com
9650-000860-01 Rev. C
- Quick Links:
- Product Description
Hide quick links:
Advertisement
Table of Contents
Troubleshooting