Table of Contents
Advertisement
Quick Links
Advertisement
Chapters
Table of Contents
Troubleshooting

Summary of Contents for ZOLL E Series
- Page 1 E Series Service Manual ® 9650-1450-01 Rev. D January 2014...
- Page 2 Service Manual (REF 9650-1450-01 Rev. D) is January, 2014. If more than 3 years have elapsed since this date, contact ZOLL Medical Corporation to determine if additional product information updates are available. Copyright © 2014 by ZOLL Medical Corporation. All rights reserved. AutoPulse, Base Power Charger, CPR-D-padz, E Series, Pedi-padz, Protocol Assist Code Markers, Real CPR Help, Rectilinear Biphasic, RescueNet, See-Thru CPR, SmartAlarms, Stat-padz, SurePower, and ZOLL, are trademarks or registered trademarks of ZOLL Medical Corporation in the United States and/or other countries.
-
Page 3: Table Of Contents
Overview ..................................1-1 Before You Begin the Maintenance Tests....................... 1-2 Equipment that You Need to Perform the E Series Maintenance Tests............... 1-3 Equipment You Need for the E Series Options Maintenance Tests ..............1-3 Physical Inspection of the Unit ..........................1-5 Front Panel Button Test ............................ - Page 4 Chapter 2 Troubleshooting Overview ..................................2-1 Troubleshooting................................ 2-2 ZOLL E Series Error Messages ..........................2-5 Chapter 3 Replacement Parts Replacement Parts..............................3-2 Parts Diagrams................................3-7 Chapter 4 Functional Description Main System Board..............................4-2 High Voltage Module..............................4-7 AC/DC Charger Module ............................4-10 System Interconnect Module ..........................4-11...
- Page 5 High Voltage Assembly Removal .......................... 5-23 Battery Interconnect Board Removal........................5-24 A/C Charger Assembly Removal ........................... 5-26 Access Detect Cable Removal ..........................5-27 Capacitor Removal ..............................5-28 Connector Panel Removal ............................. 5-29 Communication Board & PCMCIA Module Removal ................... 5-31 Recorder Removal ..............................
- Page 6 9650-1450-01 Rev.D...
- Page 7 Safety and effectiveness data submitted by ZOLL Medical Corporation to the Food and Drug Administration (FDA) under section 510(K) of the Medical Device Act to obtain approval to market is based upon the use of ZOLL accessories such as disposable electrodes, patient cables and batteries. The use of external pacing/defibrillation electrodes and adapter units from sources other than ZOLL is not recommended.
- Page 8 Do not discharge a battery pack except in a ZOLL SurePower™ Charger Station or compatible ZOLL Battery Charging/Testing unit. Do not use the E Series in the presence of flammable agents (such as gasoline), oxygen-rich atmospheres, or flammable anesthetics. Using the unit near the site of a gasoline spill may cause an explosion.
- Page 9 Purchase Order for a unit with an expired warranty. • If the unit needs to be sent to ZOLL Medical Corporation, obtain a Service Request number from the Technical Service representative. Return the unit in its original container to: 9650-1450-01 Rev. D...
- Page 10 Technical Service for International Customers International customers should return the unit in its original container to the nearest authorized ZOLL Medical Corporation Service Center. To locate an authorized service center, contact the International Sales Department at ZOLL Medical. See back cover of this manual.
-
Page 11: Chapter 1 Maintenance Tests
The E Series has two checkout procedures: the operator’s shift checklist and the extensive six-month maintenance tests checkout procedures. Because the E Series units must be maintained ready for immediate use, it is important for users to conduct the Operator’s Shift Checklist procedure at the beginning of every shift. -
Page 12: Before You Begin The Maintenance Tests
Assemble the tools or specialized testing equipment listed in the “Equipment You Need to Perform the Maintenance Tests” section shown below. • Keep an extra fully charged ZOLL E Series compatible battery available. • Schedule an hour to conduct the entire maintenance test. -
Page 13: Equipment That You Need To Perform The E Series Maintenance Tests
Be sure to follow the manufacturer’s recommendations for conducting the maintenance tests. We recommend the use of the following equipment when performing E Series Maintenance Tests: ZOLL Medical Electrode Adapter from Fluke Biomedical (ZOLL Part Number 3010-0378). - Page 14 E Series Service Manual Fluke Biomedical Cufflink Analyzer (if NIBP option is installed) • Note: The Fluke Biomedical BP Pump NIBP Monitor Analyzer and the Fluke Biomedical BP Pump NIBP Analyzer use different technologies for testing NIBP monitors and therefore, the manual provides two different procedures for performing the NIBP Volume Leak test with each of these types of test equipment.
-
Page 15: 1.0 Physical Inspection Of The Unit
E Series Service Manual 1.0 Physical Inspection of the Unit Tools Needed: Battery. Test Setup: None. Observe this... Pass/Fail/NA Housing Is the unit clean and undamaged? Does the unit show signs of excessive wear? Does the handle work properly? Does the recorder door open and close properly? - Page 16 E Series Service Manual Observe this... Pass/Fail/NA 1.15 Is the battery seated in the battery well correctly? Record your results on the Maintenance Test Checklist. 9650-1450-01 Rev. D...
-
Page 17: 2.0 Front Panel Button Test
Install the battery in the unit or connect the A/C power cord to the unit and then plug the cord into an electrical outlet. Connect the universal cable and ECG cable (3 lead, 5 lead, or 12 lead) to the ZOLL simulator, or Fluke Biomedical Impulse 4000 Analyzer (or equivalent). - Page 18 E Series Service Manual Do this... Verify that... Pass/Fail/NA Press the INC softkey. The bar graph appears on the display, indicating an increase in volume. Note: This action does not increase the volume of the unit’s audio prompts. 2.10 Press the DEC softkey.
- Page 19 E Series Service Manual Do this... Verify that... Pass/Fail/NA The unit discharges and the SHOCK button is no longer illuminated. 2.24 Press the SHOCK button. o o o A 15 second strip chart automatically prints, displaying the number of joules delivered (if equipped with recorder and configured to print event).
-
Page 20: 3.0 3, 5, And 12 Leads Test
Tools Needed: 3 lead, 5 lead, and 12 lead cables. Test each cable separately. QED 6 Defibrillator Analyzer. Test Setup: 1. The E Series unit must be configured to display ECG LEAD OFF message. Connect the lead wires appropriate to each test to the QED 6 Defibrillator Analyzer. -
Page 21: Power Supply Test (Optional)
Set the power supply voltage to 7V. Caution Be sure to connect the power supply properly to the E Series battery well terminals or damage to the unit may result. Do NOT raise the power supply voltage above 12V. 9650-1450-01 Rev. D... - Page 22 E Series Service Manual 15 Amp Battery Well Supply Black Do this... Verify that... Pass/Fail Turn the selector switch to MONITOR. The unit does not turn on. (For AED units, turn the selector switch to ON and select Manual mode.) Turn the selector switch to the OFF position.
-
Page 23: 5.0 Leakage Current Test
E Series Service Manual 5.0 Leakage Current Test Tools Needed: See the manufacturer’s instructions or supplied specifications for the leakage tester you use. Test Setup: See the manufacturer’s instructions or supplied specifications for the leakage tester you use. Repeat leakage test with accessories: MFC, external paddles, and anterior/posterior paddles. -
Page 24: Paddles Test (If Applicable)
E Series Service Manual 6.0 Paddles Test (If applicable) Tools Needed: QED 6 Defibrillator Analyzer. Test Setup: If applicable, connect the universal cable to the paddles and place the paddles in paddle wells. Do this... Verify that... Pass/Fail/NA Turn the selector switch to DEFIB. Press and The energy selection decreases to 1J. -
Page 25: 7.0 Heart Rate Display Test
E Series Service Manual 7.0 Heart Rate Display Test Tools Needed: ECG Cable (3 or 5 leads). • QED 6 Defibrillator Analyzer. • Test Setup: 1. Turn the selector switch to MONITOR. Press LEAD button until “I” displays. Connect the ECG leads to the QED 6 Defibrillator Analyzer. -
Page 26: 8.0 Calibrating Pulses On Strip Chart Test
E Series Service Manual 8.0 Calibrating Pulses on Strip Chart Test Tools Needed: None Test Setup: None Do this... Verify that... Pass/Fail/NA Press the RECORDER button. The strip chart displays a signal of 300 ppm with an amplitude of ... -
Page 27: 9.0 Notch Filter Test
E Series Service Manual 9.0 Notch Filter Test Tools Needed: QED 6 Defibrillator Analyzer. Test Setup: 1. Connect the ECG cable to the QED 6 Defibrillator Analyzer. Connect the ECG cable to the unit Do this... Verify that... Pass/Fail/NA Turn the selector switch to MONITOR mode. -
Page 28: 10.0 Heart Rate Alarm Test
E Series Service Manual 10.0 Heart Rate Alarm Test Tools Needed: QED 6 Defibrillator Analyzer Do this... Verify that... Pass Fail/NA 10.1 Turn the selector switch to MONITOR mode. The monitor screen displays the Lead II message and the waveform for... - Page 29 E Series Service Manual Do this... Verify that... Pass Fail/NA 10.14 Press and hold ALARM SUSPEND for 4 o o o seconds to disable alarms. Record your results on the Maintenance Tests Checklist. 9650-1450-01 Rev. D 1-19...
-
Page 30: 11.0 Defibrillator Self Test
E Series Service Manual 11.0 Defibrillator Self Test WARNING! SHOCK HAZARD! TAKE THE NECESSARY PRECAUTIONS TO GUARD AGAINST SHOCK OR INJURY BEFORE YOU START CONDUCTING THE DEFIBRILLATOR TESTS. Keep hands and all other objects clear of the multi-function cable connections and defibrillator analyzer when discharging the defibrillator. - Page 31 E Series Service Manual Note: The universal cable should not be connected to any equipment at the beginning of this test. Do this... Verify that... Pass/Fail 11.1 Turn the selector switch to DEFIB mode. The unit displays the CHECK PADS/POOR PAD CONTACT message.
-
Page 32: 12.0 Synchronized Cardioversion Test
E Series Service Manual 12.0 Synchronized Cardioversion Test Tools Needed: QED 6 Defibrillator Analyzer. Test Setup: 1. Connect the universal cable via the adapter (D.N.I #3010-0378) to the defibrillator analyzer. Set ECG on analyzer to 60 -120 BPM. Do this... -
Page 33: 13.0 Shock Test
1. Connect the universal cable via the adapter (D.N.I #3010-0378) to the defibrillator analyzer. Ensure that a fully charged battery is installed in the unit. Note: If your E Series AED does not have manual override capability, do not perform this test. Do this... Verify that... - Page 34 E Series Service Manual Do this... Verify that... Pass/Fail/NA 13.10 Press the CHARGE button. Wait for the The unit displays the message, DEFIB 100J RDY. o o o SHOCK button to illuminate. 13.11 Press the SHOCK button. The unit discharges 83J-122J into the simulator.
-
Page 35: 14.0 Summary Report Test
E Series Service Manual 14.0 Summary Report Test Tools Needed: QED 6 Defibrillator Analyzer. Test Setup: 1. Connect the universal cable to the QED 6 analyzer. If you are using paddles, place the paddles on the analyzer’s discharge plates. Do this... -
Page 36: 15.0 Advisory Message Test
E Series Service Manual 15.0 Advisory Message Test Tools Needed: QED 6 Defibrillator Analyzer. Test Setup: 1. Connect the universal cable via the adapter (D.N.I #3010-0378). Attach the E Series to the QED 6 Defibrillator Analyzer. Do this... Verify that... Pass/Fail 15.1 Turn the selector switch to DEFIB mode. -
Page 37: 16.0 Pacer Test
Note: The following tests are to be performed only on E Series units equipped with the optional pacing function. The pacer output can be measured using an oscilloscope set to DC coupling connected across a load resistor. (See diagram in column for universal cable connector polarity.) The load resistor is a 100 ohm, 5 watt or greater. - Page 38 Pacer rate on QED 6 analyzer is 177-183 ppm 16.9 Connect the ECG cable to the E Series and The unit displays ECG at 60 BPM with no stimulus markers. QED 6 analyzer. Select the ECG at 60 BPM on the QED 6 analyzer.
- Page 39 17.3 Press the 02+ or 02- softkey of the simulator The SpO reading of 98 +/- 1% appears on the E Series monitor. until the SpO output is at 98%. Note: You may need to wait up to 2 minutes for the information to appear on the ZOLL display.
- Page 40 The SpO simulator screen displays an SpO saturation rate of 70-74%. BPM- softkey until the heart rate is at 230 BPM. The E Series monitor displays a heart rate in the heart position of 226-234 BPM. 17.10 Remove the Masimo patient cable.
- Page 41 E Series. 18.2 Set the front panel switch to MONITOR or ON. The E Series monitor displays the WARM UP message. For AED units, enter Manual Mode. Note: Warm up may take about 1 minute. 18.3 When the WARM UP message disappears, The E Series monitor displays the ZERO DONE message.
-
Page 42: 19.0 Barometric Pressure Calibration Check
Press the Restore Defaults softkey. The difference between the current barometric pressure reading (obtained in o o o Step 19.1) and the barometric pressure reading that the E Series unit displays is <30. 19.5 Use the Inc> and Dec< softkeys to enter the o o o current barometric pressure on the unit. -
Page 43: Co2 Accuracy Check (Optional Test To Check Etco2 Sensor)
Connect the CAPNOSTAT 5 CO2 Mainstream cable with airway adapter attached, or the CAPNOSTAT 5 Sidestream cable with the cannula attached, to the yellow connector at the back of the E Series. 20.2 While pressing and holding the second softkey The unit displays EtCO Calibration screen. - Page 44 E Series Service Manual Do this... Observe this... Pass/Fail/NA 20.9 Attach a regulated flowing gas mixture of 5% The gas flow rate should already be preset to 2 to 5 liters per minute. /balance Nitrogen (N ) to the airway adapter.
-
Page 45: 21.0 Nibp Volume Leak Test With Fluke Biomedical Nibp Analyzer
Make sure the ECG cable is not connected to the E Series unit. If the SpO2 option is installed, make sure that the Masimo cable is NOT connected to the E Series unit. NIBP Volume Leak Test with Fluke Biomedical Cufflink NIBP Analyzer Do this... - Page 46 • Displays no Volume Leak reading, but maintains a stable pressure reading at or above 200 mmHg, then the E Series unit has passed the test; there is no volume leak. In addition, the E Series unit displays the simulator’s pressure reading in the “Cuff Pressure”...
-
Page 47: 22.0 Nibp Transducer Calibration Test
These instructions apply to the Fluke Biomedical BP Pump; for equivalent devices, follow the manufacturer’s instructions. Make sure the ECG cable is not connected to the E Series unit. If the SpO2 option is installed, make sure that the Masimo cable is NOT connected to the E Series unit. Do this... - Page 48 (for example, 205 mmHg). 22.8 On the E Series unit, press the Read Cuff The value that the E Series unit displays is accurate within +/- 3mmHg of the softkey when the simulator’s Measured screen pressure parameter value set on the NIBP simulator.
-
Page 49: 23.0 Nibp Monitor Test
NIBP Simulators may produce a reading on the NIBP monitor that is shifted from the simulator's setting. The offset value must be established based on a statis- tical sample of monitors and readings. Please contact ZOLL Technical Support if you require assistance establishing the offset of the simulator and test set-up that you are utilizing. - Page 50 These values only apply for test set-ups utilizing the BP Pump 2 Simulator. Variations of the test set-up or different simulators may produce readings outside the provided values and will require end-user facility to establish the appropriate offset and tolerances. Please contact ZOLL Technical Support i f you require assistance establishing the offset o f y o u r simulator and test set-up.
-
Page 51: 24.0 Bluetooth Test
24.0 Bluetooth Test Tools Needed: USB Bluetooth dongle with driver CD. Bluetooth application software BlueSoleil. Test Setup: Verify that the E Series unit is configured as follows: Bluetooth -- INSTALLED; Bluetooth Baud Rate -- 115200. Do this... Verify that... Pass/Fail 24.1... - Page 52 E Series Service Manual (This page is intentionally left blank) 1-42 9650-1450-01 Rev. D...
-
Page 53: Overview
Troubleshooting tables for ECG Leads Off Messages and Monitor Displays • ZOLL E Series Error Messages • If the problems you encounter are not listed below, call ZOLL Medical Corporation’s Technical Service Department for further assistance. (See page vii for contact information.) 9650-1450-01 Rev. D... -
Page 54: Troubleshooting
E Series Service Manual Troubleshooting The following tables show the most common troubleshooting issues and their solutions. First, attempt to solve the problem with “Recommended User Action.” If these steps do not solve the problem, follow the steps listed in the “Recommended Technical Action”... - Page 55 • Remove and replace the High Voltage Module. defibrillator from service. • Remove and replace the system board. • Call ZOLL Technical Support for assistance. Flash or arcing under • Avoiding using alcohol and betadine in and around the treatment •...
- Page 56 Same as above example. Electronic interference. Check for possible excessive radio frequency interference. • Turn off sources of excessive RFI. Move patient cables away from other electrical equipment. • Move E Series unit away from RFI source. 9650-1450-01 Rev. D...
-
Page 57: Zoll E Series Error Messages
ZOLL E Series Error Messages The following is a list of ZOLL E Series error messages that may appear on your display. The “User Advisory” column informs you about an action in progress or provides feedback on a user correctable situation that typically does not require further technical support. The “Technical Action” column describes what you as a technician can do to correct the situation. - Page 58 E Series Service Manual Error Message Explanation User Technical Action Advisory CABLE FAULT (Auto defib mode only.) Replace paddle set, universal cable and/or system board. Incorrect A/D reading for paddle ID (similar to PADDLE FAULT). CARD FULL Memory Card Full.
- Page 59 User prompt issued simultaneously with other faults if Possible configuration problem. Replace high defib is disabled. voltage module. Call ZOLL Technical Support. DEFIB FAULT 71 More than 50 internal dumps occurred in less than 20 Turn the unit to OFF and back on. If fault persists, minutes.
- Page 60 E Series Service Manual Error Message Explanation User Technical Action Advisory DEFIB FAULT 109 Defib capacitor voltage is greater than selected energy Replace high voltage module or capacitor. when defibrillator is charging or ready. DEFIB FAULT 111 Defib capacitor voltage has exceeded the absolute Replace high voltage module, capacitor, and or maximum acceptable voltage.
- Page 61 E Series Service Manual Error Message Explanation User Technical Action Advisory ECG LEAD OFF One or more ECG leads are not properly connected when Check cable and patient connection. Change leads are selected as input. electrodes. Prepare patient’s skin.
- Page 62 E Series Service Manual Error Message Explanation User Technical Action Advisory INSERT CARD No card installed in unit during manual or semi-automated modes. Check memory card LOW BATTERY Low battery. Replace battery or plug into AC power. Replace charger.
- Page 63 E Series Service Manual Error Message Explanation User Technical Action Advisory PADDLE FAULT Cannot detect type of accessory attached to the universal Replace paddles, internal paddles, system board, cable. high voltage module and/or universal cable. PERFORM CPR Advisory message in AED auto defib mode.
- Page 64 E Series Service Manual Error Message Explanation User Technical Action Advisory REPLACE BATTERY Battery voltage is less than absolute minimum. Shutdown Replace with charged battery. imminent. REPLACE CARD Write errors during manual or semi-automated modes. May have configuration card installed or write protection on.
- Page 65 E Series Service Manual Error Message Explanation User Technical Action Advisory COMM ERR No transmissions from SpO unit received. Communication Replace Sp0 module and/or system board. error or no communication from Sp0 module. PULSE SEARCH Pulse search in progress.
- Page 66 E Series Service Manual Error Message Explanation User Technical Action Advisory USER SETUP REQ Both copies of stored cal/config data are bad or have Perform configuration setup. never been programmed. VF ALARMS OFF VF alarms disabled in pace mode or when paddles are selected as leads.
-
Page 67: Replacement Parts
This section contains a listing of the replacement parts available for the ZOLL E Series devices. Replacement parts may be ordered through an authorized ZOLL distributor or directly from ZOLL Medical Corporation. The prices for parts are available from ZOLL Medical Corporation’s Technical Service Department. - Page 68 E Series Service Manual Replacement Parts Item numbers correspond to the parts diagrams beginning on page 3-7. Description Part Number Item # UPPER HOUSING, E SERIES 1005‐0026 SUBASSY, PRINTER, E SERIES 1005‐0001 SCR,#6‐32,MCH,1‐7/8",PNH,PHL,SST, RoHS 0163‐0454 SCR,#4‐40,MCH,1/4",PNH,PHL,STL,ZNC,EXT, RoHS 0163‐0900 LABEL SET, SERIAL NUMBER, W/BAR CODE 9305‐0513 WASHER, EXT, #6, SS, RoHS 0163‐0027 CABLE, SYSTEM INTERCONNECT TO PRINTER 9500‐0725 INTERCONNECT, E SERIES MAG.TOROID.FERRITE.HT3.81MM OD8.51MM ID3.45MM 0140‐0106 CABLE, BATTERY INTERCONNECT TO SYSTEM ...
- Page 69 E Series Service Manual Description Part Number Item # ASSY, PCB, COMMUNICATION CARRIER, W/BLUETOOTH, 9301‐0390‐02 E SERIES SCR.PPH.#4 X.0.375"ZI.HI/LO 0163‐1217 CABLE, SYSTEM INTERCONNECT TO COMMUNICATIONS 9500‐0724 CARRIER, E SERIES SUBASSY, AC CHARGER, E SERIES 1005‐0003 ASSY, E SERIES, HV MODULE 1001‐0278‐02 HVM BASE, ESERIES 9310‐0904 CABLE, BATTERY INTERCONNECT TO HVM, DEFIB , E SERIES 9500‐0720 CABLE, BATTERY INTERCONNECT TO HVM, E SERIES 9500‐0718 TAPE KAPTON, 1 ¼" WIDE, 0.0025" THICK 36 YDS LONG 0550‐0003 ASSY, CONNECTOR PANEL, AC, E SERIES 1005‐0021‐03 SCR.PPH.#6.x 3/4".STL.ZN.HI/LO 0163‐0409 CLAMP, HOUSING, CO, E SERIES 9310‐0981 ASSY, CAPACITOR, AC CHARGER, SEQ E020, SUB ASSY 1, 1005‐0024 E SERIES TY‐RAP CABLE TIE STD 18 LB .093W X 3.62 L 0501‐0001 CABLE, MFC SIGNAL, TO HV MODULE, E SERIES...
- Page 70 E Series Service Manual Description Part Number Item # ASSY, MID FRAME, GENERIC, E SERIES 1005‐0005‐01 SCR,#4‐40,MCH,1/2",PNH,PHL,SST,EXT, RoHS 0163‐0410 TYWRAP CABLE .070"Wx.030"x2.8"LG NYLON 0501‐0023 MX‐1 BOARD 3005‐0005 SCR.PPH.4 X.0.250"..ZI.HI/LO 0163‐1215 ASSY, PCB, ESERIES, ISOLATED POWER SUPPLY 9301‐1383‐01 CABLE, FLEX, SYSTEM TO ISOLATED POWER SUPPLY, E SERIES 9500‐0779 CABLE, FLEX, MX‐1 TO ISO POWER SUPPLY, E SERIES 9500‐0786 STOP, CONNECTOR HEADER, E SERIES 9330‐0484 TAPE KAPTON, 1 ¼" WIDE, 0.0025" THICK 36 YDS LONG 0550‐0003 NIBP ADV2 MODULE W/PUMP, E SERIES 1005‐0018 ASSY, LOWER HOUSING, E SERIES 1005‐0014 CABLE, AC RECEPTACLE TO CHARGER 9500‐0739 SCR,#4‐40,MCH,7/16",FLT,PHL,SST, RoHS 0163‐0779 SCR.PPH.6X.0.625"..ZI.HI/LO 0163‐1232 ASSY, INTERLOCK CONNECTOR, E SERIES 1005‐000035‐01...
- Page 71 E Series Service Manual Description Part Number Item # CABLE, CONTROL TO DISPLAY, POWER 9500‐0712 CABLE, DF9, CONTROLS TO SYSTEM PWBA, E SERIES 9500‐0776 ASSY, DISPLAY BACKER, PLATE, E SERIES 1005‐0010 GASKET, BEEPER, M‐SERIES 9161‐0010 WASHER, ANTI‐ROTATION 9160‐0012 CUP, PACER 9310‐0519 WASHER, ANTI‐ROTATION 9160‐0011 DIFFUSER, REVIVER 9310‐0590 PANEL, FRONT, PUSHBUTTON, REVIVER 9310‐0549 ISOPROPANOL.REAGENT GRADE.99.8% PURITY 0550‐0186 SUPPORT, CONTROL BOARD, E SERIES 9310‐0932 SNAP RIVET BLACK NYLON 0163‐1709 LABEL, SUB‐ASSY, BAR CODE ID 0305‐0020 BEZEL, TOP, E SERIES 9310‐1913 SCR,#6‐32,MCH,1/4",PNH,PHL,STL,ZNC,INT, RoHS 0163‐0278 COVER, PCMCIA, E SERIES 9310‐0914...
- Page 72 E Series Service Manual Description Part Number Item # ASSY, CABLE, MULTI FUNCTION AND TEST PORT 1001‐0194‐01 GREASE CHEMICAL RESISTANT 0550‐0500 ASSY, HANDLE, E SERIES 1005‐0028 WASHER 0.010 IN SHIM.FOR SHDR SCR.0.251 IN ID X.0.375 0163‐0452 IN OD.18‐8 SST. SCR.SKT HD/HEX.10‐32.0.250".303 SS.0.1880" SHLD LEN 0163‐0406 W/PATCH KNOB, PACER 9310‐0520 SEAL, KNOB, INJECTION MOLDED, M‐SERIES 9310‐0698 KNOB, MAIN W/SPRING, M/E SERIES 9310‐0704‐01 FILLER, LI‐ION BATTERY, ESERIES 9310‐0903 SCR,#6‐32,MCH,1/2",PNH,PHL,SST,EXT, RoHS 0163‐0918 LABEL, PANEL CONNECTOR, NIBP/ETCO2, AC, LEFT, E SERIES 9305‐0931‐01 LABEL, PANEL CONNECTOR, SPO2, E SERIES 9305‐0938‐01 LABEL, PANEL CONNECTOR, MFC/ECG, RIGHT, E SERIES 9305‐0937‐01 No Options Label SCR,#4‐40,MCH,1/4",PNH,PHL,STL,ZNC,EXT, RoHS 0163‐0900 BRACKET, PCMCIA MODEM LOCK...
-
Page 73: Parts Diagrams
E Series Service Manual Parts Diagrams 9650-1450-01 Rev. D... - Page 74 E Series Service Manual 9650-1450-01 Rev. D...
- Page 75 E Series Service Manual 9650-1450-01 Rev. D...
- Page 76 E Series Service Manual 3-10 9650-1450-01 Rev. D...
- Page 77 E Series Service Manual 9650-1450-01 Rev. D 3-11...
- Page 78 E Series Service Manual 3-12 9650-1450-01 Rev. D...
- Page 79 E Series Service Manual 9650-1450-01 Rev. D 3-13...
- Page 80 E Series Service Manual 3-14 9650-1450-01 Rev. D...
- Page 81 E Series Service Manual 9650-1450-01 Rev. D 3-15...
- Page 82 E Series Service Manual 3-16 9650-1450-01 Rev. D...
- Page 83 E Series Service Manual 9650-1450-01 Rev. D 3-17...
- Page 84 E Series Service Manual 3-18 9650-1450-01 Rev. D...
- Page 85 E Series Service Manual 9650-1450-01 Rev. D 3-19...
- Page 86 E Series Service Manual 3-20 9650-1450-01 Rev. D...
- Page 87 E Series Service Manual 9650-1450-01 Rev. D 3-21...
- Page 88 E Series Service Manual 3-22 9650-1450-01 Rev. D...
- Page 89 E Series Service Manual 9650-1450-01 Rev. D 3-23...
- Page 90 E Series Service Manual 3-24 9650-1450-01 Rev. D...
- Page 91 E Series Service Manual 9650-1450-01 Rev. D 3-25...
- Page 92 E Series Service Manual 3-26 9650-1450-01 Rev. D...
- Page 93 E Series Service Manual 9650-1450-01 Rev. D 3-27...
- Page 94 E Series Service Manual 3-28 9650-1450-01 Rev. D...
- Page 95 E Series Service Manual 9650-1450-01 Rev. D 3-29...
- Page 96 E Series Service Manual 3-30 9650-1450-01 Rev. D...
-
Page 97: Chapter 4 Functional Description
E Series Service Manual Chapter 4 Functional Description This chapter provides functional descriptions of the components contained in the ZOLL E Series and the E Series options. Refer to the interconnect diagram that delineates the different components of the defibrillator. This chapter includes: Main System Board •... -
Page 98: Main System Board
Main System Board The E Series electrical circuitry consists of several functional modules. Each module is physically located on one or more of the printed wiring board assemblies (PWBA). In some cases, a functional module is distributed across several assemblies within the unit. The main components of the E Series... - Page 99 E Series Service Manual Refer to the E Series Interconnect diagram to identify unit components described in this manual. Module Location Main Central Processing Unit (CPU) and ECU Main System PWBA Pacer/Defib Charging and High Voltage Control (Defib/Pace) High Voltage Module...
- Page 100 Main System Board Functions The Main System Board contains the major computing and control elements for the E Series unit. The printed wiring board assembly (PWBA) receives signals from the front panel control switches, ECG input connectors and functional modules, such as the AC/DC charger, pacer/defibrillator modules,...
-
Page 101: Power Supply
Power Supply The power supply converts DC power from a removable battery or the AC/DC Battery Charger module to voltages required by the E Series hardware. The power supply circuit converts the raw battery or the Charger PWBA output voltages of +8.5 VDC to +16 VDC into the voltages shown in the table below, including load and line regulation. - Page 102 E Series Service Manual ECG Front End The ECG front end provides an electrically isolated serial interface between the main system board functions and patient interface ECG connectors. It performs the following: Analog ECG amplification and signal conditioning. • Pacemaker pulse detection.
-
Page 103: High Voltage Module
E Series Service Manual High Voltage Module The High Voltage (HV) module includes the high voltage circuitry required for pacing and defibrillation, including the defib charge circuitry, patient relay, safety relay, defib capacitor, defib choke and front end protection circuitry for the MFC ECG. - Page 104 When the patient discharge SCRs are deactivated, the safety relay closes to internally dissipate any remaining energy. If the E Series is in the self test mode, the energy is delivered internally. The microprocessor calculates the actual delivered energy from the current waveform and displays a TEST OK message on the display, if the self test meets the appropriate criteria.
- Page 105 E Series Service Manual The patient relay discharges via the signal PATREL_DRV generated by XPATREL and Q304, Q323, and Q322. PATREL_DRV is disabled when XPACE_SEL is at a logic low. When the patient relay activation completes, the Main System Board releases the XPATREL signal. Several hundred milliseconds later, the safety relay closes to ensure the high voltage capacitor energy is completely dissipated.
-
Page 106: Ac/Dc Charger Module
(for a mains input), AC-DC and DC-DC conversion and isolation barrier between the E Series and power sources. This module also provides the power necessary to run the E Series in any mode of operation, as well as providing additional charging current to the battery. When the E Series is turned off but connected to an external AC or DC source, the charger module controls battery charging currents and voltages needed to charge the E Series battery. -
Page 107: System Interconnect Module
E Series Service Manual System Interconnect Module The system interconnect PWBA receives signals from the Main System Board and in turn controls operation of the stripchart recorder and PCMCIA functions. Stripchart Recorder The Stripchart Recorder module includes a microprocessor, serial interface to the main system board and circuitry which drive the stripchart recorder’s motor and printhead in response to the main CPU signals. -
Page 108: E Series Options
The 12 lead cable is required to produce 12 lead reports. E Series unit must have the 12 lead option installed. All limb leads and V-leads must be connected to initiate a 12 lead acquisition. Printed 12 Lead bandwidth is user configurable to be either 0.05-150 Hz (per AAMI EC11) or 0.05-40 Hz. The 0.05-40 Hz bandwidth selection is used to reduce noise artifact in the high end of the diagnostic frequency range. - Page 109 Circuit Board) connects to the Masimo sensors and reports monitoring results (oxygen saturation, pulse rate, pulse waveform, etc.) via a serial digital interface to the E Series system board. The E Series system provides isolated DC power and serial communication to the SpO Board via the Isolated Power Supply board.
- Page 110 The NIBP option is designed to measure blood pressure for adult and pediatric patients. The patient blood pressure cuff and hose connect to the E Series unit through the NIBP connector on the rear panel of the unit. The NIBP button on the front panel of the E Series allows you to initiate and terminate blood pressure measurements.
- Page 111 E Series Service Manual Chapter 5 Disassembly Procedures This chapter provides instructions on how to disassemble and reassemble the E Series unit, and includes the following sections: “Front Panel Removal” on page 3 “Front Panel Disassembly” on page 5 “Bottom Panel Removal” on page 10 “NIBP Pump Removal”...
-
Page 112: Required Equipment
E Series Service Manual Required Equipment #1 Philips head screwdriver • #2 Philips head screwdriver • #24 Hex head screwdriver • Wooden stick • Needlenose pliers • Wire Cutters • Exactor Knife • 1/2” Nut Driver • Safety Precautions Warning! - Page 113 E Series Service Manual Front Panel Removal 1. Remove handle by removing the screw and washer on each 2. Remove the 3 screws on the top of the panel. side. 3. Front panel assembly slides down. Use caution when removing 4.
- Page 114 E Series Service Manual 5. Remove the clear plastic spacer from between the cable 6. Disconnect the cable from the system board by pulling connector and upper housing by grasping the center with straight away from the board. needlenose pliers, and pushing straight up.
-
Page 115: Front Panel Disassembly
E Series Service Manual Front Panel Disassembly There are 3 styles of front panel assemblies. (Original style has 1 cutout in back plate, the 2nd release had 2 cutouts, and the latest release Note: has 2 cutouts along with a slot for the display invertor.) Original Style 2nd Style Latest Style Slot 1. Remove 8 screws from backing plate (Both Styles). 2. Lift backing plate out of the frame by prying from the top (Both styles). - Page 116 E Series Service Manual 3. 2 Cutout Style: Remove black rivet by pulling it straight up. 3A. Original Style: Remove 2 screws, and lift out shield. Then remove plastic isolator. 4. Remove 3 cables from the control board. 5. Remove display by lifting from the bottom.
- Page 117 E Series Service Manual 6. Flip front panel over. Remove main selector knob by gently 7. Remove 1/2” nut from the main selector knob, then remove lifting. (Note: Older version may be glued and need to be pried the anti-rotation washers.
- Page 118 E Series Service Manual 10. Cut video cable tie wrap. 11. Push on board retaining clip to release the control board from front panel frame, but do not remove board until next step. Note: When reinstalling see Table 5.1 at end of section for setting control board dip switches.
- Page 119 E Series Service Manual 13. Using an exacto knife pry corner of front panel membrane up, 14. Using an exacto knife pry corner of data entry switch up, and remove by gently pulling off the membrane. and remove by gently pulling off the membrane.
-
Page 120: Bottom Panel Removal
E Series Service Manual Bottom Panel Removal 1. Remove the 9 screws securing the base. 2. Remove bottom panel by lifting panel straight up. 5-10 9650-1450-01 Rev. D... -
Page 121: Nibp Pump Removal
E Series Service Manual NIBP Pump Removal 1. Cut tie wrap, and disconnect hose from the NIBP fitting. 2. Remove the screw that is holding the NIBP Pump. Note: When reinstalling, do not over tighten tie wrap. 3. Disconnect ECG flex cable from system board, by releasing 4. - Page 122 E Series Service Manual 5. Carefully lift and remove pump assembly. Note: When reinstalling, lift flex cable so power connector on NIBP board is under the cable. 5-12 9650-1450-01 Rev. D...
-
Page 123: System Board Removal
E Series Service Manual System Board Removal 1. Remove plastic spacer by gently lifting. 2. Remove PCMCIA short connecting ribbon cable from both sides. Note: During reassembly verify the spacer ridge sits on board. 3. Remove flat UBP cable from both ends by releasing jams on 4. - Page 124 E Series Service Manual 5. Remove gray printer ribbon cable straight out of the board. 6. Remove colored stranded cable by pulling cable downward Gently lift system board straight up half way from the casing. from the connector. Remove the black stranded cable.
- Page 125 E Series Service Manual 9. From the opposite corner, remove colored stranded cable. 10. Gently separate the system board from its cradle. Board has tabs holding it into slots in the metal frame Note: at the bottom. 11. Remove the 3 flat cables by pulling connector straight out, and remove system board.
-
Page 126: System Interconnect Board Removal
E Series Service Manual System Interconnect Board Removal 1. Remove the 2 screws holding the interconnect board to the 2. Remove board, carefully twist flat cable and feed through frame. plastic support to remove frame. Note: Fold cable over in 45 degree angle as shown below. - Page 127 E Series Service Manual O2 Board Removal (MS-11) 1. Remove the 2 flat cables from the SPO2 board by pulling 2. Remove the 4 screws from each corner of the SPO2 board. straight away from board. Remove the SPO2 board from the device.
-
Page 128: Isolated Power Supply Removal
E Series Service Manual Isolated Power Supply Removal 1. Disconnect the molex connector. 2. Disconnect the 2 spade connectors. 3. Remove the 4 screws securing the board. 4a (Old Style MS-11) Carefully lift the isolated power supply and remove long grey stranded cable (EtCO2), and the 2 flat cables, then remove board. - Page 129 E Series Service Manual 4b (New Style MX-1) Carefully lift the isolated power supply and remove short grey stranded cable (EtCO2), and the 2 flat cables, then remove board. 9650-1450-01 Rev. D 5-19...
-
Page 130: Bridge Board Removal
E Series Service Manual Bridge Board Removal 1. Remove high voltage isolator (clear) by removing tape and then 2. Remove MFC retainer (if present). rotating around MFC signal cable. 3. Remove MFC socket from the chassis and wire loom. Cut tie 4. - Page 131 E Series Service Manual 5. Loosen the 3 screws holding plastic frame in place. Lift the 6. Remove bridge board isolator (clear). entire frame out of the chassis. 7. Disconnect flex cable from bridge board by lifting the tabs and 8.
- Page 132 E Series Service Manual 9. Remove bridge board from the mid-frame by carefully guiding cable through the slot. 5-22 9650-1450-01 Rev. D...
-
Page 133: High Voltage Assembly Removal
E Series Service Manual High Voltage Assembly Removal 1. Disconnect small flat cable by lifting up jams on each side and 2. Disconnect J104 cable by lifting up jams on each side and pull straight out, then disconnect larger cable by pulling straight pull straight out. -
Page 134: Battery Interconnect Board Removal
E Series Service Manual Battery Interconnect Board Removal 1. Remove the 3 machine screws from the battery pins (J100, 2. Remove the 2 screws securing the board. J101, J102). 3. Gently remove board by lifting from this end. 4. Disconnect flat cable by pulling gently. - Page 135 E Series Service Manual 5. Remove 3 ferrite rings from the battery pins, and pull pins straight out. 9650-1450-01 Rev. D 5-25...
-
Page 136: A/C Charger Assembly Removal
E Series Service Manual A/C Charger Assembly Removal 1. Remove cables from cable harnesses, and disconnect tie wrap 2. Remove screw from A/C ground cable. on the MFC signal cable. 3. Disconnect A/C connector cable by pulling down on latch and pull towards you. -
Page 137: Access Detect Cable Removal
E Series Service Manual Access Detect Cable Removal 1. Push the 4 pin connector through the connector panel 2. Disconnect ferrite from looms, and remove cable from squeezing the tabs. Remove nut holding ECG Out/RS232 device. connector, and push connector through panel. -
Page 138: Capacitor Removal
E Series Service Manual Capacitor Removal WARNING! This unit may contain lethal voltages. You MUST completely discharge the high voltage capacitor before removing from unit. DO NOT SHORT THE TERMINAL ENDS OF THE CAPACITOR. 1. Lift capacitor out. 2. Bleed the excess voltage using a resistor with values of approximately 5 kohms, 25 watts for 10-20 seconds. -
Page 139: Connector Panel Removal
Connector Panel Removal Note: There are 2 styles of SPO2 connector (MS-11 & MX-1) used on the E Series, use the appropriate instructions for disassembly. 1. (MS-11 Style) Remove the 2 screws securing the panel to the 2. Lift assembly out, gently sliding rubber ground sleeve off housing. - Page 140 E Series Service Manual 1. (MX-1 Style) Peel back SPO2 label using an exacto knife. 2. Remove 2 screws and gently lift up on connector panel. 3. Remove the 2 screws securing the panel to the housing. 4. Remove rubber gasket (black foam) from connector panel.
- Page 141 E Series Service Manual Communication Board & PCMCIA Module Removal 1. Remove the 4 screws securing the communication board to the 2. Remove the screw that is securing the PCMCIA board to housing. the housing. Lift PCMCIA assembly straight out.
-
Page 142: Recorder Removal
E Series Service Manual Recorder Removal 1. Lift up on the PCMCIA cover, and remove large screw securing 2. Open printer door, then lift bezel straight out. bezel to housing. 3. The recorder assembly can be removed by loosening 4 screws 4. - Page 143 E Series Service Manual 5. Pivot the roller housing back and it will come off. 6. Remove the two washers securing the printer interconnect board by gently prying up on them. Remove the board. 7. Slide pivot pin out of printer frame.
- Page 144 E Series Service Manual 9. The print head can removed by lifting out of the frame. 5-34 9650-1450-01 Rev. D...
-
Page 145: Appendix A Overview
Interconnect Diagram for the E Series Unit • ZOLL E Series Maintenance Tests Checklist • Photocopy the checklists and use the copies to record the results of the maintenance tests performed on the E Series equipment; keep them for your records. 9650-1450-01 Rev. D... - Page 146 Printer 9500- 9301-0390-01 Motor 0724 Communications 9500-0723-50 9500-0736 9500-0729 NIBP Assembly Print Head 9301-0385-01 * - integrated assembly Paper/Recorder Sensors ** - assembly in tegration planned Hose Rev 0.01 Figure 1: Interconnect Diagram for E Series Unit 9650-1450-01 Rev. D...
- Page 147 ZOLL E Series Serial No. Location Maintenance Test Checklist Tester Signature Date 1.0 Physical Inspection 2.0 Front Panel Button 3.0 Leads Test 7.0 Heart Rate Display 11.0 Defibrillator Test Test Self Test Result of Check: Pass Fail Pass Fail...
- Page 148 ZOLL E Series Serial No. Location Maintenance Test Checklist Tester Signature Date 13.0 Shock Test 15.0 Advisory Message 17.0 SpO Monitor Test 20.0 CO Accuracy 24.0 Bluetooth Test Test Check (Optional) Pass Fail Pass Fail Pass Fail Pass Fail 13.2...
- Page 149 E Series Operator’s Guide ® 9650-1210-01 Rev. R...
- Page 150 The issue date for the E Series Operator's Guide (REF 9650-1210-01 Rev. R) is March, 2013. If more than 3 years have elapsed since this date, contact ZOLL Medical Corporation to determine if additional product information updates are available. Copyright © 2013 by ZOLL Medical Corporation. All rights reserved. AutoPulse, Base Power Charger,...
- Page 151 FDA Regulations ....................1-15 Tracking Requirements ..................1-15 Notification of Adverse Events ................1-15 Software License....................1-15 Service ......................... 1-15 Returning a unit for service ................1-15 The ZOLL Serial Number ..................1-16 9650-1210-01 Rev. R E Series Operator’s Guide...
- Page 152 E Series Operator’s Guide SECTION 2 OPERATING CONTROLS AND INDICATORS ........2-1 Code Markers ......................2-4 Summary Report Function..................2-4 Summary Report Formats..................2-4 Defibrillation Format.....................2-5 Pacer Format (Pacer version only) ..............2-6 Heart Rate Alarm Activated Format ..............2-7 VF Alarm Activated Format (Refer to Section 6)..........2-7 Recorder On Format ....................2-7...
- Page 153 5-Lead Monitoring....................10-5 Changing from 3-Lead Monitoring ..............10-5 Changing from 5-Lead ECG Monitoring.............10-5 Simultaneous 3-Lead Printing ................10-5 Vital Signs Trending ....................10-5 Viewing Vital Signs Trending Data on the Display..........10-5 Printing a Vital Signs Trend Report..............10-6 9650-1210-01 Rev. R E Series Operator’s Guide...
- Page 154 Manual Method ....................11-5 Automated Dial-up Method ................11-5 GPS Synchronization..................11-6 Operator’s Shift Checklist for E Series Products (Manual)......11-7 Operator’s Shift Checklist for E Series Products (Semiautomatic) ....11-8 SECTION 12 BATTERY MANAGEMENT ............... 12-1 Battery Care......................12-1 Battery Life Expectancy ..................12-1 LOW BATTERY Message..................12-1...
- Page 155 Guidance and Manufacturer’s Declaration — Electromagnetic Emissions ..A-7 Electromagnetic Immunity Declaration (EID) ............. A-8 EID for Life-Support Functions ................A-9 Recommended Separation Distances from RF Equipment for E Series Life-Support Functions ..................A-10 EID for Non-Life-Support Functions ..............A-11 Recommended Separation Distances from RF Equipment for E Series Non-Life-Support Functions ................
- Page 156 (This page intentionally left blank.)
-
Page 157: General Information
(DC) mains and an easily replaced battery pack that is quickly recharged in the device when it is connected to AC or DC mains. In addition, the unit’s batteries may be recharged and tested using ZOLL Base Power Charger™ 4X4 or ZOLL SurePower™... -
Page 158: How To Use This Manual
How to Use This Manual The E Series Operator's Guide provides information operators need to know for the safe and effective use and care of the E Series products. Before operating this device, be sure to read and understand all the information contained within. - Page 159 PCMCIA Data Cards (2 per package) 8000-0551 (8Mb) 8000-0552 (16Mb) 8000-0553 (32Mb) RS232 Data Transfer Cable 8000-0605-01 ECG Simulator 8012-0206 * The terms “ZOLL Multi-Function Electrode (MFE) Pads” and “MFE Pads” are used interchangeably throughout this manual. 9650-1210-01 Rev. R...
-
Page 160: Symbols Used On The Equipment
E Series Operator’s Guide Symbols Used on the Equipment Any or all of the following symbols may be used in this manual or on this equipment: Type B equipment. Type BF equipment. Type CF equipment. Defibrillator-proof type BF equipment. Defibrillator-proof type CF equipment. - Page 161 General Information Conformité Européenne Complies with the medical device directive 93/42/EEC. Contains lead. Recycle or dispose of properly. Keep away from open flame and high heat. Do not open, disassemble, or intentionally damage. Do not crush. Nonrechargeable battery Do not discard in trash. Recycle or dispose of properly. Date of manufacture.
- Page 162 E Series Operator’s Guide Manufacturer. Authorized representative in the European Community. Serial Number. Catalogue number. Consult instructions for use. Protected against ingress of solid foreign objects > 2.5 mm in diameter. IP34 Protected against splashing water. 9650-1210-01 Rev. R...
-
Page 163: Defibrillator Function
In Manual mode, the E Series unit may also be used for analyzing the ECG. If you are using the device in an synchronized cardioversion to terminate atrial fibrillation... -
Page 164: External Pacemaker Function (Pacer Version Only)
(asystole) ensues, you should use the The unique design of the E Series products allow clear pacemaker. viewing and interpretation of the electrocardiogram Ventricular or supraventricular tachycardias may be... -
Page 165: Pediatric Pacing
Poor adherence and/or air under the MFE Pads can Multi-Function Electrode (MFE) Pads. lead to the possibility of arcing and skin burns. The pacer version of the E Series paces using ZOLL MFE Pads. 3. Open the pad packaging and apply one edge of the pad securely to the patient. -
Page 166: Monitor
CHARGER ON indicators remain extinguished. If your lead selections — I, II, III, aVR, aVL, aVF, V (with • E Series unit does not function as expected, refer to the ECG cable), PADDLES, or PADS (APLS if connected “Troubleshooting Guidelines” on page 13-1. -
Page 167: Safety Considerations
General Information Safety Considerations The E Series products are high energy devices capable of delivering up to 200 joules. To completely deactivate the device, you must turn the selector switch to the OFF position. • In order to disarm a charged defibrillator, do one of the following: Turn the selector switch to MONITOR, OFF or... -
Page 168: Operator Safety
The ECG out signal is delayed by up to 25 ms. This delay must be considered when the ECG out signal is used as an input to other devices requiring R-wave synchronization. The E Series device may not perform to specifications when stored at the upper or lower extreme limits of storage •... -
Page 169: Cautions
General Information The AutoPulse Plus is only intended for use on adults 18 years of age or older. When using the E Series and • AutoPulse Plus as a system, this age restriction applies to the E Series as well. -
Page 170: Restarting The Device
E Series Operator’s Guide Restarting the Device Certain events require the E Series products to be restarted after they shut off or become inoperative. One example is when the battery runs down and the unit shuts off. In this case, perform these steps in this order: 1. -
Page 171: Fda Regulations
Fax: (978) 421-0010 Returning a unit for service Tel: (978) 421-9655 Before sending a unit to the ZOLL Technical Service Notification of Adverse Events Department for repair, obtain a service request (SR) Under the Safe Medical Devices Act (SMDA), health number from the service representative. -
Page 172: The Zoll Serial Number
• A product serial number of six or more alphanumeric characters The product code for the E Series defibrillator is AB. The first two characters of the date-of-manufacture code give the last two digits of the year (for example, “06”... -
Page 173: Operating Controls And Indicators
SECTION 2 OPERATING CONTROLS AND INDICATORS 11 12 13 17 18 ZM-04-36 Rev. A 1. Selector Switch Allows selection of OFF, MONITOR, DEFIB, and PACER, (Pacer version only) modes. 2. ENERGY SELECT Buttons Allows selection of defibrillation energy level. There are two sets of up-down arrow buttons; one set located on the front panel and the other (not shown) located on the sternum paddle. - Page 174 DEFIB or MONITOR mode and MFE Pads 13. CHARGER ON Indicators or Paddles are connected to the Multi-Function cable. When the E Series unit is plugged into AC mains, the Lead II is automatically selected when the E Series unit CHARGER ON indicators operate as described powers up in PACER mode (Pacer version only).
- Page 175 21. Microphone (Optional) Located on top of the unit, the recorder compartment Records audio activity in the vicinity of the E Series unit holds the paper supply for the recorder. Open the cover for storage on the PCMCIA data card.
-
Page 176: Code Markers
When you press the CODE MARKER button on the front panel, the unit displays a preconfigured list of clinical Note: Diagnostic bandwidth recordings are not included actions. See the E Series Configuration Guide (part in the summary report function. number 9650-1201-01) for information on configuring Summary Report records each event in chronological code markers. -
Page 177: Defibrillation Format
Operating Controls and Indicators as well as space for patient name, date and comments. On the last event recorded, “SUMMARY COMPLETE” is printed at the bottom left of the recorder strip. Defibrillation Format The summary report function records 6 seconds of pre-shock and 9 seconds of post-shock patient ECG data. Also recorded are joules selected, joules delivered, sync if active (includes sync markers), ECG lead, ECG size, patient impedance, actual event time and date. -
Page 178: Pacer Format (Pacer Version Only)
E Series Operator’s Guide Pacer Format (Pacer version only) The summary report function records 6 seconds of pre-pacer patient ECG data. Also recorded are the ECG lead, ECG size, patient's heart rate, actual event time and date. The date/time printed on the top of the strip corresponds to the ECG data occurring 6 seconds before the event. -
Page 179: Heart Rate Alarm Activated Format
Operating Controls and Indicators Heart Rate Alarm Activated Format The summary report function records 6 seconds of pre-alarm patient ECG. Also recorded are the ECG lead, ECG size, patient's heart rate, actual event time, and date. The date/time printed on the top of the strip corresponds to the ECG data occurring 6 seconds before the event. -
Page 180: Analyze Format
E Series Operator’s Guide and pacing current are also recorded. If async pace is active, the annotation “ASYNC PACE” is recorded. AED units additionally include shock count and AED mode annotations. Actual event date/time Analyze Format The summary report function records six seconds of pre analysis ECG and 12 seconds of ECG recorded during the ECG analysis interval with the annotation “SHOCK ADVISED”... -
Page 181: Manual Mode Activated
3. Use the scroll keys on top of the unit to scroll through the list of start times associated with different calls. AED versions of the E Series will record a “MANUAL MODE STARTED” summary report event when the device is switched from Semiautomatic mode to Manual... -
Page 182: Adding A Patient Name And Id Number To A Report
E Series Operator’s Guide Manual mode started (AED only) • Select the Enter ( ) key from the keypad and • press the Commit ( ) key on top of the unit. • Alarms triggered • Code markers The highlight automatically advances to the Recorder turned on Patient ID# line. -
Page 183: Modifying A Patient Name And Id Number
Operating Controls and Indicators Note: You cannot add a patient name to summary 5. When you have modified the patient name, move the report events already stored in memory. The cursor to the Enter ( ) key on the keypad and press patient name is stored only with those summary the Commit ( ) key on top of the unit. - Page 184 E Series Operator’s Guide (This page intentionally left blank.)
-
Page 185: Manual Defibrillation
The AutoPulse Plus is only intended for use on adults 18 years of age or older. • When using the E Series and AutoPulse Plus as a system, this age restriction applies to the E Series as well. You can perform manual defibrillation using either paddles or MFE pads;... - Page 186 The ENERGY INCREMENTED message appears when this occurs. This function is disabled if you manually change the energy level outside the pre- programmed sequence and deliver a shock. See the E Series Configuration Guide for more details. 9650-1210-01 Rev. R...
- Page 187 Manual Defibrillation Charge Defibrillator Deliver SHOCK Press the CHARGE button on the front panel (if using WARNING MFE pads) or on the apex paddle handle (if using • Warn all persons in attendance of the patient to paddles). STAND CLEAR prior to defibrillator discharge. Do not touch the bed, patient, or any equipment •...
-
Page 188: Troubleshooting
Paddle plates and handles must be thoroughly cleaned after each use. See “General Maintenance” on page 11- 1 for correct cleaning procedure. Troubleshooting If your E Series unit does not function as expected, see“Troubleshooting Guidelines” on page 13-1. 9650-1210-01 Rev. R... -
Page 189: Advisory Defibrillation
• adults 18 years of age or older. When using the Begin CPR following medical protocols E Series and AutoPulse Plus as a system, this age Request additional assistance. restriction applies to the E Series as well. Prepare the patient and attach the MFE pads With Advisory defibrillation, the unit analyzes the This is described in “MFE Pad Application/Connection”... - Page 190 CPR or other cardiopulmonary life support and re- shock. See the “Energy Level” sections (for Shock 1, analyze the ECG at appropriate intervals. Shock 2, and Shock 3) of the E Series Configuration Guide for more details. When a shockable rhythm is detected (ventricular fibrillation or tachycardia with heart rate >...
-
Page 191: Shock Advised
On/Off probability that the current ECG rhythm will be The E Series unit can be configured to use the Shock successfully converted by an immediate defibrillation Conversion Estimator for as many as the first four ECG shock. -
Page 192: Advisory Function Messages
Note: Reanalysis of the ECG rhythm, either manually or The prompt persists as long as a shockable rhythm is automatically (see E Series Configuration Guide), being detected. Press the ANALYZE button to begin is inhibited for 3 seconds after a shock. -
Page 193: Troubleshooting
This voice prompt is issued only if MFE pads were previously connected to the patient. Troubleshooting If your E Series unit does not function as expected, see the Defibrillator Troubleshooting section starting on page 13-5. 9650-1210-01 Rev. R... - Page 194 E Series Operator’s Guide (This page intentionally left blank.)
-
Page 195: Automated External Defibrillator (Aed) Operation
18 years of age or older. When using the resumes operation and displays and announces a E Series and AutoPulse Plus as a system, this age CHECK PATIENT prompt if a shockable rhythm is restriction applies to the E Series as well. -
Page 196: Aed Semiautomatic Operation
9650-0720-01) for instructions on properly configured a different energy level, you may select it by connecting the E Series to the AutoPulse Plus. It is using the ENERGY SELECT arrow buttons. The new recommended that the user cycle through the lead energy setting is displayed on the monitor. -
Page 197: Operating Messages
3 seconds after a shock. defibrillator is charged and ready once a decision Continue Patient Care to shock has been made. The E Series will not automatically disarm the defibrillator if the Continue patient care according to medical protocols. - Page 198 E Series Operator’s Guide ready for operation. Additional tone signals are ECG TOO LARGE/RETRY ANALYSIS • described below. These messages appear when the ECG signal is too large for proper rhythm analysis. Press the The messages that appear on the monitor depend upon ANALYZE button again to begin ECG analysis.
-
Page 199: Aed Manual Mode Operation
• “NonInvasive Temporary Pacing (Pacer Version Only)” on page 9-1 “ECG Monitoring” on page 10-1 • Troubleshooting If your E Series unit does not function as expected, see the “Troubleshooting Guidelines” on page 13-1. 120J SEL. 00:01 Manual Param Mode... - Page 200 E Series Operator’s Guide (This page intentionally left blank.)
-
Page 201: Synchronized Cardioversion
Prior to attempting synchronized cardioversion, ensure that the ECG signal quality is sufficient to minimize risk • of synchronizing on artifact. Synchronized cardioversion is disabled when the E Series unit is connected to the AutoPulse Plus and the • AutoPulse Plus is compressing. - Page 202 E Series Operator’s Guide Turn Selector Switch to MONITOR Press the SYNC softkey again to reactivate SYNC mode. Changing the selected energy does not turn off Sync mode. You can configure the unit to stay in Sync mode after defibrillation if desired.
-
Page 203: Troubleshooting
The charge ready tone then stops and the defibrillator remains in SYNC mode. Troubleshooting If your E Series unit does not function as expected, see the Defibrillator Troubleshooting section starting on page 13-5. 9650-1210-01 Rev. R... - Page 204 E Series Operator’s Guide (This page intentionally left blank.)
-
Page 205: Real Cpr Help
The Real CPR Help function is not intended for use on patients under 8 years of age. The CPRD-to-MFC connector is intended for use with the E Series, or other ZOLL defibrillators where indicated. • Real CPR Help is disabled when using CPR-D-padz through an AutoPulse Plus. -
Page 206: Cpr Compressions Indicator
(fully release) their hands from the patient’s chest after rate or depth, the E Series will display the letter R for compressions to allow full recoil. Rate and/or the letter D for depth to assist the rescuer in... -
Page 207: Displaying The Cpr Compressions Bar Graph
Wave 2 softkey until the bar graph is displayed. mmHg CPR Dashboard Whenever CPR-D-padz or CPR stat-padz are connected to the E Series unit and the CPR Dashboard is configured on, the unit illuminates the CPR Dashboard, Depth Rate replacing the right half of the Wave 2 display area. - Page 208 E Series Operator’s Guide (This page intentionally left blank.)
-
Page 209: See-Thru Cpr
SECTION 8 SEE-THRU CPR WARNING • The See-Thru CPR filter works only when the E Series defibrillator is monitoring CPR in Manual mode. The See-Thru CPR filter stops if: • — Diagnostic bandwidth mode is active. — The unit is in pace mode. - Page 210 E Series Operator’s Guide The following figure shows a patient in Fine VF. It is difficult for a rescuer to discern this rhythm during CPR compressions. When the CPR filter turns on, the Fine VF rhythm becomes more obvious. FineVF...
- Page 211 See-Thru CPR The following figure shows a patient in VF, which, during compressions, is slightly more difficult to discern. When viewing this ECG, it is possible to view the underlying rhythm as the filter is able to reject all of the CPR artifact. CoarseVF Raw ECG Raw ECG...
- Page 212 E Series Operator’s Guide The following figure shows a patient in PEA, which could easily be mistaken for Fine VF because enough of the compression artifact leaks through to distort this signal. When the CPR filter turns on, the PEA is still not obvious because of the left over ripples from the CPR signal.
- Page 213 See-Thru CPR The following figure shows a patient with an organized rhythm where See-Thru CPR effectively filters out artifact created by CPR. SinusRhythm Raw ECG Raw ECG • | Filter ON Filtered ECG Filtered ECG 0:00 0:12 SinusRhythm Raw ECG Raw ECG •...
- Page 214 E Series Operator’s Guide (This page intentionally left blank.)
-
Page 215: Noninvasive Temporary Pacing (Pacer Version Only)
• Pacing must be turned off before defibrillating with a second defibrillator. Otherwise, the E Series unit may be damaged. Noninvasive Temporary Pacing Attach and connect the MFE pads This is described in “MFE Pad Application/Connection”... - Page 216 E Series Operator’s Guide The pacer rate increments or decrements by a value of Electrical Capture 2 ppm on the display when you turn the knob. Electrical capture means that the unit is delivering sufficient electrical current to stimulate the heart as seen on the ECG trace.
-
Page 217: Special Pacing Applications
NonInvasive Temporary Pacing (Pacer Version Only) by increasing the pacer output (mA) until every pacing • The MFE cable is defective. marker is immediately followed by a wide QRS complex. • MFE Pads are not connected to the Multi-Function cable. Note that changing ECG leads and size can sometimes MFE Pads are not making good skin contact. -
Page 218: Asynchronous Pacing
Carefully follow all instructions provided on electrode package. Troubleshooting If your E Series unit does not function as expected, see the troubleshooting section “Pacer (Pacer version only)” on page 13-4. 9650-1210-01 Rev. R... -
Page 219: Section 10 Ecg Monitoring
ECG leads are a defibrillation-protected Type CF patient connection. Introduction IEC Color AHA Color Placement of Electrodes You can use the E Series products for either short-term Coding Coding or long-term ECG monitoring. E Series products have built-in protection circuitry to... -
Page 220: Monitoring Electrodes Attachment
E Series Operator’s Guide Monitoring Electrodes Attachment Attach snap-on leads to electrodes and check for good contact between the electrode and the lead termination. Peel the protective backing from the ECG electrode. Be careful to keep adhesive surface free of electrolyte gel. -
Page 221: Alarms
) if available. preset at 30 bpm (bradycardia) and 150 bpm (tachycardia). Refer to the E Series Configuration Guide The E Series unit has three levels of alarms as follows: for details on setting power-up alarm limits. • High Priority — Reflects physiological parameters To set alarms: that are out of bounds. -
Page 222: Suspending And Silencing Alarms
Deactivating and Activating Alarms The unit restores the original high heart rate alarm limit when ECG monitoring resumes. To deactivate all alarms on the E Series unit, press and hold down the ALARM SUSPEND button for 3 seconds Suspending and Silencing Alarms or longer. -
Page 223: 5-Lead Monitoring
ECG Monitoring standard monitoring bandwidth when you release the set to Yes in System Configuration. Refer to the E Series RECORDER button. Configuration Guide for more information. 5-Lead Monitoring Vital Signs Trending You can perform 5-lead ECG monitoring with the Some E Series units include a vital signs trending feature appropriate ECG patient cable. -
Page 224: Printing A Vital Signs Trend Report
E Series Operator’s Guide If only one parameter is installed on the unit, the trend Data is printed on the stripchart in order of newest data screen automatically appears when you press the Trend to oldest data. Alarm conditions are indicated on the softkey. -
Page 225: Clearing Vital Signs Trend Records
ECG Monitoring Clearing Vital Signs Trend Records The unit stores at least 24 hours of normal (non-alarmed, non-NIBP) trending samples before it is overwritten. If the unit is powered down while recording trend data, the corresponding gap in time is indicated on the display by a series of asterisks in the time field and no data points in any parameter fields. - Page 226 E Series Operator’s Guide (This page intentionally left blank.)
-
Page 227: Section 11 General Maintenance
1. Open the recorder cover on top of the E Series unit. Operator's Shift Checklists are included at the end of this 2. Remove the paper (if necessary). -
Page 228: Testing The Defibrillator (Semiautomatic Mode)
SHOCK button and that it displays and announces 1. Verify adult paddles are installed and are inserted all the PRESS SHOCK prompt. the way into their holders on the side of the E Series 12. Press and hold the SHOCK button and verify unit unit. -
Page 229: Testing The Pacer (Pacer Version Only)
4. Verify that the units displays the TEST OK message and prints a stripchart of the event, noting the energy delivered and impedance data. Note: If TEST FAILED appears, contact the ZOLL Technical Service Department immediately. Testing the Pacer (Pacer Version Only) Perform these tests on all pacer-equipped units periodically. -
Page 230: Changing Recorder Paper
4. Align the paper above the open tray. The proper orientation is with the black arrows pointing up, and the word “ZOLL Medical Corporation” running along the left side, as shown. 5. Slide the paper into the tray. -
Page 231: Setting Time And Date
General Maintenance Setting Time and Date Note: The last field does not automatically scroll (wrap) to the beginning. You must press the Prev Field Check the time and date on the recorder annotation. If it softkey to enter the values for the last field. is not correct, reset the time and date (from System If you need to make corrections, press the Prev Utilities mode) manually, by dial-up to a National Institute... -
Page 232: Gps Synchronization
The modem could not dial the phone ERROR number. Ensure that the modem card is properly connected. Ensure that the user-selected dial prefix is correct. (Refer to E Series Configuration Guide for information). Retry. NIST DATA The unit detected an error in the data ERROR from NIST. -
Page 233: Operator's Shift Checklist For E Series Products (Manual)
Operator’s Shift Checklist for E Series Products (Manual) Recommended checks and procedures to be performed at the start of each shift. For more detailed information, see the E Series Operator’s Guide. Date____________________________Location________________________________Unit Serial Number__________________ 1. Condition Remarks Shift Shift Shift Unit clean, no spills, clear of objects on top, case intact 2. -
Page 234: Operator's Shift Checklist For E Series Products (Semiautomatic)
Operator’s Shift Checklist for E Series Products (Semiautomatic) Recommended checks and procedures to be performed at the start of each shift. For more detailed information, see the E Series Operator’s Guide. Date_____________________________Location __________________________Unit Serial Number ______________________ 1. Condition Remarks Shift... -
Page 235: Section 12 Battery Management
Changing the Battery Pack operation, and the pattern of discharging and recharging batteries contribute to the loss of battery charge capacity. The E Series products are designed for quick removal Because of this, ZOLL recommends that operators and replacement of the battery pack. -
Page 236: Charging And Testing Battery Packs
ZOLL Base Power Charger 4x4 or SurePower Charger was designed specifically for this purpose. With the E Series unit plugged in to AC mains and turned off, the device recharges the sealed lead acid battery to greater than 90% capacity within 4 hours, and the lithium-ion battery pack to greater than 90% capacity in less than 7 hours. -
Page 237: Achieving Optimal Battery Pack Performance
It is important to visibly distinguish battery packs that are charged from those that are not. Establish a system for visually indicating whether a battery pack is charged and ready for use or is in need of charging. ZOLL can provide you with battery pack status labels for this purpose, or you can use labels or methods of your own. - Page 238 If the device shows a LOW BATTERY warning during testing, replace the depleted battery pack with a fully charged one, and recharge the depleted battery pack. DON’T charge battery packs at temperature extremes. ZOLL recommends charging battery packs at or near normal room temperature (15°C to 35°C or 59°F to 95°F). 12-4 9650-1210-01 Rev. R...
-
Page 239: Section 13 Troubleshooting Guidelines
This section addresses many of the common problems or questions that may arise during operation. If trouble persists after consulting this guide, contact the appropriate technical personnel or ZOLL Technical Service Department. A more detailed troubleshooting guide is found in the E Series Service Manual. - Page 240 AutoPulse Plus, if in use, and plug the multifunction cable directly into the electrode pads. Noisy ECG, artifact, wandering baseline • Consider 1 – 21Hz filter bandwidth (see E Series Configuration Guide). Prepare the patient’s skin prior to electrode attachment. •...
-
Page 241: Recorder
Recorder makes stuttering sound when activated. • Check recorder for paper jam. Light or poor quality printing is observed. • Ensure correct paper type (ZOLL P/N 8000-0300) is in use. • Ensure paper is installed grid-side against recorder print head. -
Page 242: Pacer (Pacer Version Only)
E Series Operator’s Guide Pacer (Pacer version only) Symptom Recommended Action Unit displays the CHECK PADS prompt. • Ensure MFE Pads are connected to Multi-Function cable. Ensure electrode gel is not dry. Replace MFE Pads if • necessary. • Ensure good electrode-to-patient contact. -
Page 243: Defibrillator
Troubleshooting Guidelines Defibrillator Symptom Recommended Action Excessive artifact when using paddles as ECG source. • Ensure PADDLES is selected. Firmly press paddles against patient skin. • Use gel on paddles. • • Clean paddle surface. • Check and clean between adult and pediatric shoe. Check cable for damage. - Page 244 Unit displays the DEFIB MAINT. REQUIRED message. • Contact ZOLL Technical Service Department. Unit displays a DEFIB FAULT XX message. If problem persists, contact ZOLL Technical Service • Department. 13-6 9650-1210-01 Rev. R...
-
Page 245: Ac Charger
Troubleshooting Guidelines AC Charger Symptom Recommended Action The green and orange-yellow CHARGER ON indicators Verify battery is installed. • illuminate alternately. Turn unit ON to identify the fault condition. • • Replace battery pack with a fully charged battery pack. If problem persists, replace battery pack, unplug device •... - Page 246 E Series Operator’s Guide (This page intentionally left blank.)
-
Page 247: Appendix Aspecifications
Operating: 0C to 55C (32F to 131F) Storage Temperature: -20 to 60C (-4F to 140F) Note: The E Series device may not perform to specifications when stored at the upper or lower extreme limits of storage temperature and immediately put into use. -
Page 248: Pacemaker (Pacer Version
Variable from 30 ppm to 180 ppm ±1.5% (increments or decrements by a value of 2 ppm) Output protection Fully defibrillator protected and isolated Multi-Function Specifically designed adult anterior/posterior pre-gelled ZOLL MFE Pads and Electrode (MFE) Multi-Function stat-padz packaged in pairs Pads... -
Page 249: Ecg Monitoring
0.1ms and 2ms, without overshoot, and between ±100mV and ±500mV, with widths between 0.1ms and 2ms, with overshoot from 4 to 100ms. The pacemaker pulse rejection capability for the E Series with pacemaker pulses and a normally paced QRS and T wave includes pulses between ±2mV and ±700mV amplitude, with widths between 0.1ms and 2ms, without overshoot, and between ±2mV... -
Page 250: Cpr Monitoring
The E Series averages the interval between the last 5 detected beats. On startup, the Averaging E Series averages the rate between detected beats once two beats are detected, until a full 5 beats have been received. The rate is updated every beat. After this condition is met, the meter is updated every beat with an average of the last 5 beats. -
Page 251: Display
Display Screen Type High resolution, color liquid crystal display (LCD) Screen Size 5.63 inches (14.3 cm) diagonally Sweep Speed 25 mm/s Viewing Time 4 seconds Messages ERASING REPORT, INSERT CARD, CARD FULL, REPLACE BATTERY, LOW BATTERY, PERFORM CPR, ECG TOO LARGE, NOISY ECG, RETRY ANALYSIS, CHECK PATIENT, ANALYSIS HALTED, PRESS ANALYZE, NO SHOCK ADV., CHECK PULSE, SHOCK ADVISED, PRESS CHARGE, SELECT PADS, SELECT ECG LEADS, SELECT DEFIB MODE, VF ALARMS OFF, DISABLE SYNC, ANALYSIS RESTARTED,... - Page 252 Standby Life: one month before retest and recharge. Note: Each monitoring option added to the E Series device decreases the Operating Time that can be obtained from a fully charged battery. Refer to the individual option insert for the operating run time specific to your device. For further details specific to your device, contact the ZOLL Technical Service Department.
-
Page 253: Guidance And Manufacturer's Declaration - Electromagnetic Emissions
Guidance and Manufacturer’s Declaration Electromagnetic Emissions — The E Series unit is intended for use in the electromagnetic environment specified below. Ensure that the E Series unit is used in such an environment. Emissions Test Compliance Electromagnetic Environment — Guidance... -
Page 254: Electromagnetic Immunity Declaration (Eid
If the user 40% U (60% dip in U 40% U (60% dip in U supply input lines of the E Series unit requires for 5 cycles for 5 cycles IEC 61000-4-11 continued operation during 70% U (30% dip in U... -
Page 255: Eid For Life-Support Functions
To assess the electromagnetic environment due to fixed RF transmitters, an electromagnetic site survey should be considered. If the measured field strength in the location in which the E Series unit is used exceeds the applicable RF compliance level above, the E Series unit should be observed to verify normal operation. If abnormal performance is observed, additional measures may be necessary, such as reorienting or relocating the E Series unit. -
Page 256: Recommended Separation Distances From Rf Equipment For E Series Life-Support Functions
The life-support functions on the E Series unit are defined to be any function associated with Pacing and Defibrilla- tion. Specifically, these functions include, but are not limited to, the pacing pulse output and defibrillation energy dis- charge. -
Page 257: Eid For Non-Life-Support Functions
To assess the electromagnetic environment due to fixed RF transmitters, an electromagnetic site survey should be considered. If the measured field strength in the location in which the E Series unit is used exceeds the applicable RF compliance level above, the E Series unit should be observed to verify normal operation. If abnormal performance is observed, additional measures may be necessary, such as reorienting or relocating the E Series unit. -
Page 258: Recommended Separation Distances From Rf Equipment For E Series Non-Life-Support Functions
The non-life-support functions on the E Series unit are defined to be any function not listed as a life-support function in the “EID for Life Support Functions” table (Note a). Specifically, these functions are noninvasive blood pressure... -
Page 259: Rectilinear Biphasic Waveform Characteristics
(VT) defibrillation study. This study (which was conducted using ZOLL M Series defibrillators) and the findings are described below. Since the E Series unit's rectilinear biphasic waveform employs the same first and second phase timing, the same first and second phase currents/voltages and essentially the same mechanisms for controlling defibrillation waveshape, the ZOLL M Series and ZOLL E Series defibrillation waveforms are considered equivalent. - Page 260 ±15% Figures A-1 through A-20 show the rectilinear biphasic waveforms that are produced when the E Series defibrillator is discharged into loads of 25, 50, 75, 100, 125, 150, and 175 ohms at each energy setting (200, 150, 120, 100, 85, 70, 50, 30, 20, 15, 10, 9, 8, 7, 6, 5, 4, 3, 2 and 1 joule[s]).
- Page 261 25Ω 50Ω 75Ω 100Ω 125Ω 150Ω 175Ω Figure A-1. Rectilinear Biphasic Waveforms at 200 Joules 25Ω 50Ω 75Ω 100Ω 125Ω 150Ω 175Ω Figure A-2. Rectilinear Biphasic Waveforms at 150 Joules 25Ω 50Ω 75Ω 100Ω 125Ω 150Ω 175Ω Figure A-3. Rectilinear Biphasic Waveforms at 120 Joules 9650-1210-01 Rev.
- Page 262 E Series Operator’s Guide 25Ω 50Ω 75Ω 100Ω 125Ω 150Ω 175Ω Figure A-4. Rectilinear Biphasic Waveforms at 100 Joules 25Ω 50Ω 75Ω 100Ω 125Ω 150Ω 175Ω Figure A-5. Rectilinear Biphasic Waveforms at 85 Joules 25Ω 50Ω 75Ω 100Ω 125Ω 150Ω...
- Page 263 25Ω 50Ω 75Ω 100Ω 125Ω 150Ω 175Ω Figure A-7. Rectilinear Biphasic Waveforms at 50 Joules 25Ω 50Ω 75Ω 100Ω 125Ω 150Ω 175Ω Figure A-8. Rectilinear Biphasic Waveforms at 30 Joules 25Ω 50Ω 75Ω 100Ω 125Ω 150Ω 175Ω Figure A-9. Rectilinear Biphasic Waveforms at 20 Joules 9650-1210-01 Rev.
- Page 264 E Series Operator’s Guide 25Ω 50Ω 75Ω 100Ω 125Ω 150Ω 175Ω Figure A-10. Rectilinear Biphasic Waveforms at 15 Joules 25Ω 50Ω 75Ω 100Ω 125Ω 150Ω 175Ω Figure A-11. Rectilinear Biphasic Waveforms at 10 Joules 25Ω 50Ω 75Ω 100Ω 125Ω 150Ω...
- Page 265 25Ω 50Ω 75Ω 100Ω 125Ω 150Ω 175Ω Figure A-13. Rectilinear Biphasic Waveforms at 8 Joules 25Ω 50Ω 75Ω 100Ω 125Ω 150Ω 175Ω Figure A-14. Rectilinear Biphasic Waveforms at 7 Joules 25Ω 50Ω 75Ω 100Ω 125Ω 150Ω 175Ω Figure A-15. Rectilinear Biphasic Waveforms at 6 Joules 9650-1210-01 Rev.
- Page 266 E Series Operator’s Guide 25Ω 50Ω 75Ω 100Ω 125Ω 150Ω 175Ω Figure A-16. Rectilinear Biphasic Waveforms at 5 Joules 25Ω 50Ω 75Ω 100Ω 125Ω 150Ω 175Ω Figure A-17. Rectilinear Biphasic Waveforms at 4 Joules 25Ω 50Ω 75Ω 100Ω 125Ω 150Ω...
-
Page 267: Clinical Trial Results For The Biphasic Waveform
Figure A-20. Rectilinear Biphasic Waveforms at 1 Joules Clinical Trial Results for the Biphasic Waveform The efficacy of ZOLL’s Rectilinear Biphasic waveform has been clinically verified during a study of defibrillation of Ventricular Fibrillation (VF)/Ventricular Tachycardia (VT). A feasibility study was performed initially for defibrillation of VF/VT (n=20) on two separate groups of patients to ensure waveform safety and energy selection. -
Page 268: Randomized Multi-Center Clinical Trial For Cardioversion Of Atrial Fibrillation (Af
A total of 173 patients entered the study. Seven (7) patients who did not satisfy all protocol criteria were excluded from the analysis. ZOLL disposable gel electrodes with surface areas of 78 cm (anterior) and 113 cm (posterior) were used exclusively for the study. -
Page 269: Synchronized Cardioversion Of Atrial Fibrillation
between the two waveforms were considered statistically significant when the 95% confidence interval between the two waveforms was greater than 0%. Results: The study population of 165 patients had a mean age of 66±12 years with 116 male patients. The total efficacy of consecutive rectilinear biphasic shocks was significantly greater than that of monophasic shocks. The following table displays the Kaplan-Meier (product-limit) survival curves for each of the two waveforms. -
Page 270: Shock Conversion Estimator
SCE Level settings (HIGH, MEDIUM, and LOW) and corresponding SPI thresholds, sensitivities, and specificities that can be configured on the E Series unit. Column 1 is the SPI threshold in mv-Hz. Columns 2 and 3 are the sensitivity and specificity as described below (expressed in percent). - Page 271 Number of ECG Rhythms with SPI > Threshold that were successfully converted Sensitivity = ------------------------------------------------------------------------------------------------------------- Total number of ECG rhythms that were successfully converted Number of ECG rhythms with SPI Threshold that did not convert Specificity = ----------------------------------------------------------------------------------------- Total number of ECG rhythms that did not convert Figure A-21.
- Page 272 E Series Operator’s Guide Table A-2. Accuracy Table of SCE Levels and Corresponding SPI Thresholds SCE Level SPI Threshold Sensitivity Specificity (mV-Hz) HIGH MEDIUM 10.8 References: [1] Eftestol T, Sunde K, Steen PA. Effects of Interrupting Precordial Compressions on the Calculated Probability of Defibrillation Success during Out-of-Hospital Cardiac Arrest.
-
Page 273: Ecg Analysis Algorithm Accuracy
The data in the following table summarizes the accuracy of the ECG analysis algorithm as tested Clinical Performance Results against ZOLL’s ECG Rhythm Database. The performance of the incorporated analysis algorithm The algorithm sequence takes approximately 9 seconds in a single analysis sequence satisfies the applicable... - Page 274 E Series Operator’s Guide (This page intentionally left blank.)
-
Page 275: Appendix B Medical Report Capability
5. Press the Enter softkey. You can store up to two hours of incident data (ECG and The E Series unit erases the card and displays the unit status) or up to 38 minutes of incident data and ERASING CARD message. A progress bar appears simultaneous audio recording on one 4-megabyte while erasing the card. -
Page 276: Transferring Data To A Pc With A Pcmcia Data Card Reader
Some E Series units include a wireless communication remote handheld device or PC to access any data option that uses Bluetooth technology to communicate transmitted from the E Series unit. Refer to the ZOLL with a ZOLL-approved, Bluetooth-equipped host system Data Relay user documentation for installation and (handheld device or PC running ZOLL Data Relay operating instructions. -
Page 277: Communication Settings
You must configure the same communication settings on Upload both the E Series unit and the PC or hand-held device for proper data transmission. The correct communication settings are: [008] 12 FEB 10 06:25:37... -
Page 278: Activating Automatic Data Transmission
Param Wave 2 Alarms 12 Lead In realtime mode, the E Series unit only transmits trend data collected since the unit was powered-up, and does MONITOR NOT transmit all trend data stored in memory. To transmit all trend data, you must initiate transmission... -
Page 279: Transmitting 12-Lead Patient Records
Phone# softkey. If you are transmitting via Bluetooth, then the green 1. With the E Series unit in Monitor mode (ON for AED power LED on the E Series unit illuminates. You must units), press the 12 Lead softkey to access the 12 now establish a connection between the host system Lead menu. - Page 280 The unit operates, but no data is Wait for E Series events to clear being stored. and retry. Erasing the card may allow return Ensure remote device is on and to normal data storage operation ready to accept data.
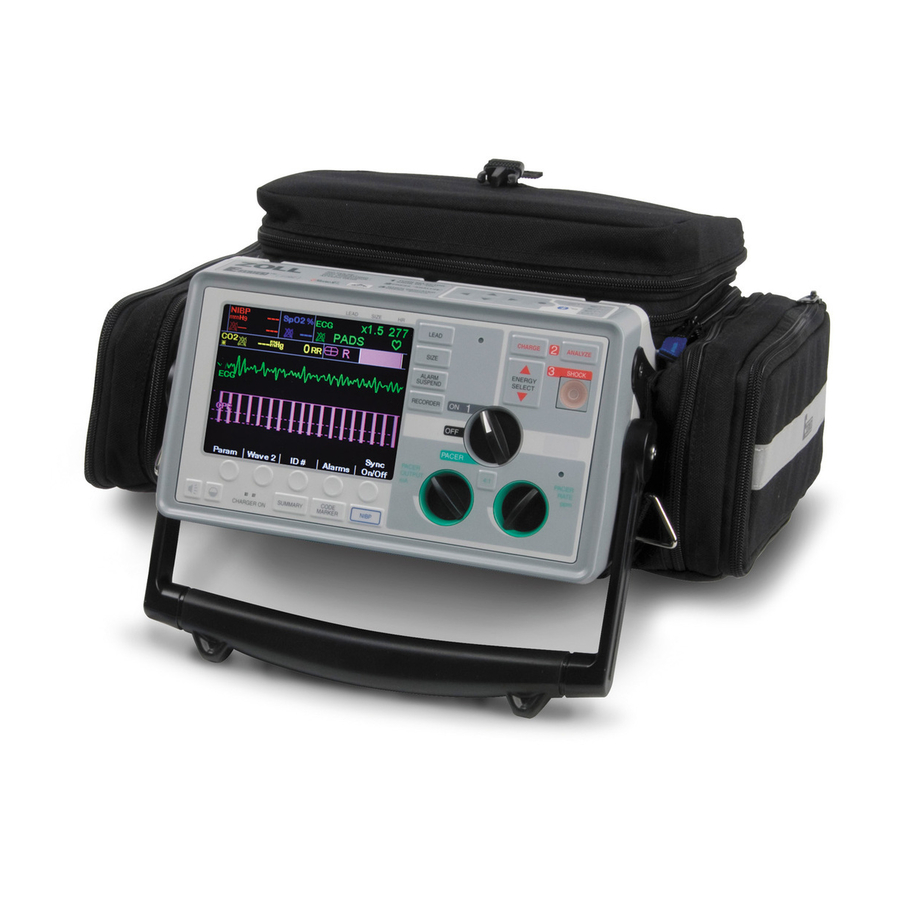















Need help?
Do you have a question about the E Series and is the answer not in the manual?
Questions and answers