Table of Contents
Advertisement
Quick Links
PROFESSIONAL MEDICAL PRODUCTS
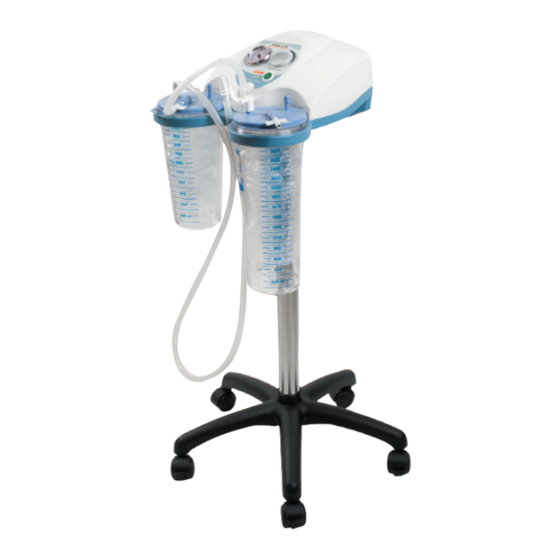
ASPIRATORE SUPERVEGA BATTERY - SU CARRELLO
SUPERVEGA BATTERY SUCTION ASPIRATOR -
ON TROLLEY
ASPIRATEUR SUPERVEGA BATTERY - SUR CHARIOT
ABSAUGER SUPERVEGA BATTERY - AUF TROLLEY
ASPIRADOR SUPERVEGA BATTERY - SOBRE CARRO
28193
Gima S.p.A.
Via Marconi, 1 - 20060 Gessate (MI) Italy
gima@gimaitaly.com - export@gimaitaly.com
www.gimaitaly.com
Made in Italy
0476
Advertisement
Table of Contents

Summary of Contents for Gima SUPERVEGA 28193
- Page 1 SUPERVEGA BATTERY SUCTION ASPIRATOR - ON TROLLEY ASPIRATEUR SUPERVEGA BATTERY - SUR CHARIOT ABSAUGER SUPERVEGA BATTERY - AUF TROLLEY ASPIRADOR SUPERVEGA BATTERY - SOBRE CARRO 28193 0476 Gima S.p.A. Via Marconi, 1 - 20060 Gessate (MI) Italy gima@gimaitaly.com - export@gimaitaly.com www.gimaitaly.com Made in Italy...
-
Page 2: Important Safety Rules
RF communication devices (mobile phones, transceivers, etc.) that may interference with the said device. 7. WARNING: Do not change this equipment without the permission of the manufacturer GIMA S.p.A. None of electric or mechanical parts has been designed to be repaired by customers or end-users. Don’t open... -
Page 3: Technical Characteristics
ENGLISH the device, do not mishandle the electric / mechanical parts. Always contact technical assistance 8. Using the device in environmental conditions different than those indicated in this manual may harm seri- ously the safety and the technical characteristics of the same. 9. -
Page 4: Cleaning Of The Device
ENGLISH Working Condition Room Temperature: 5 ÷ 35°C Room Humidity Percentage: 30 ÷ 75% RH Atmospheric pressure: 800 ÷ 1060 hPa Altitude: 0 ÷ 2000m s.l.m. Conservation condition and Transport Room Temperature: - 40°C ÷ 70°C Room Humidity Percentage: 10 ÷ 100% RH Atmospheric pressure: 500 ÷... -
Page 5: Cleaning Of Accessories
ENGLISH Service life of the device: more than 1000 hours of operation (or 3 years) in accordance with the standard conditions of testing and operation. Shelf life: maximum 5 years from the date of manufacture. WARNING: The medical device is provided without a specific suction probe. If this device must be used with a specific suction probe, the end user is responsible for making sure it complies with the EN 10079-1 regulation. - Page 6 The device is made up of a lead battery which cannot be accessed from the outside. In order to replace it, consult the technical staff authorised by the manufacturer. USE ONLY THE RECOMMENDED BATTERIES FROM GIMA. THE USE OF OTHER BATTERIES IS NOT RECOMMENDED AND INVOLVES THE CANCELLATION OF WARRANTY In the event that the service personnel has to replace the internal battery, pay special attention to the polarity of the same component.
- Page 7 2° case – If both the security system doesn’t work, there is the possibility that liquid comes inside the device, in this case return the device to GIMA technical service. GIMA S.p.A. will provide upon request electric diagrams, components list, descriptions, setting in- structions and any other information that can help the technical assistance staff for product repair.
- Page 8 ENGLISH Filter assembling Make sure the filter is assembled with the arrows on the side of the patient. WARNING: The inside of the medical device must be regularly checked for the presence of liquids or other visible contamination (secretions). In the presence of liquids or other visible contamination, immediately replace the medical device due to the risk of an insufficient vacuum flow rate.
- Page 9 ENGLISH NEVER USE THE DEVICE WITHOUT JAR AND / OR PROTECTION FILTER Using FLOVAC® disposable collection system: Before connecting the disposable collection system, remove the blu ring fitted on the tank holder for a more comfortable insertion of the same container. •...
- Page 10 ENGLISH Guidance and manufacturer’s declaration – electromagnetic Emissions The SUPERVEGA BATTERY SUCTION UNIT is intended for use in the electromagnetic environment specified below. The customers or the user of the SUPERVEGA BATTERY SUCTION UNIT should make sure that it’s used in such an environment.
- Page 11 ENGLISH Guidance and manufacturer’s declaration – Immunity Emissions The SUPERVEGA BATTERY SUCTION UNIT is intended for use in the electromagnetic environment specified below. The customers or the user of the SUPERVEGA BATTERY SUCTION UNIT should make sure that it’s used in such an environment.
- Page 12 ENGLISH Recommended separation distance between portable and mobile radio-communication devices and the monitor The SUPERVEGA BATTERY SUCTION UNIT surgical aspirator is intended to operate in an electro-magnetic environment where RF irradiated interferences are under control. The client or operator of the SUPERVEGA BATTERY SUCTION UNIT device can help prevent electro-magnetic interference by keeping a minimum distance between the portable and mobile RF communication devices (transmitters) and the SUPERVEGA BATTERY SUCTION UNIT device, as recommended below, in relation to the radio-communication maximum output power.
- Page 13 For more detailed information about recycling of this product or battery, please contact your local Civic Office, your household waste disposal service or the shop where you purchased the product. GIMA WARRANTY TERMS The Gima 12-month standard B2B warranty applies.
















Need help?
Do you have a question about the SUPERVEGA 28193 and is the answer not in the manual?
Questions and answers