Table of Contents
Advertisement
New Product Information Sheet
Specifications subject to change
© OMRON HEALTHCARE EUROPE B.V.
Model (code):
RS7 Intelli IT (HEM-6232T-E)
OMRON HEALTHCARE EUROPE B.V.
Scorpius 33 2132 LR Hoofddorp The Netherlands
P.O. Box 2050 2130 GL Hoofddorp The Netherlands
Phone: +31 (0)23 55 44 700 fax +31 (0)23 55 44 701
www.omron.healthcare.com
BANKERS JP Morgan Chase Bank NA ACC. NR.
PM-1771-E-01-03/2018
413.90.366
IBAN GB04CHAS60924241390366 SWIFT CHASGB2L
CHAMBER OF COMMERCE NR. 342.10306
BTW/VAT NR. NL8132.86.281.B01
Advertisement
Table of Contents

Summary of Contents for Omron RS7 Intelli IT
- Page 1 New Product Information Sheet Specifications subject to change © OMRON HEALTHCARE EUROPE B.V. Model (code): RS7 Intelli IT (HEM-6232T-E) OMRON HEALTHCARE EUROPE B.V. Scorpius 33 2132 LR Hoofddorp The Netherlands P.O. Box 2050 2130 GL Hoofddorp The Netherlands Phone: +31 (0)23 55 44 700 fax +31 (0)23 55 44 701 www.omron.healthcare.com...
-
Page 2: Product Description
OMRON HEALTHCARE Co., Ltd 53, Kunotsubo, Terado-cho, Muko, KYOTO, 617-0002 Japan PRODUCION FACILITY OMRON HEALTHCARE MANUFACTURING VIETNAM CO., LTD. No.28 VSIP II, Street2, Vietnam-Singapore Industrial Park II, Binh Duong Industry-Services Urban Complex, Hoa Phu Ward, Thu Dau Mot City, Binh Duong Province, Vietnam EU REPRESENTATIVE OMRON HEALTHCARE EUROPE B.V. -
Page 3: General Description
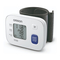
Included accessories / optional accessories General description The RS7 Intelli IT is an automatic wrist blood pressure monitor, operating on the oscillometric principle. This blood pressure monitor is to be worn on your wrist. When the cuff inflates, this monitor senses the pressure pulsations of the artery underneath the cuff. -
Page 4: Intended User
This device is a digital monitor intended for use in measuring blood pressure and pulse rate in adult patient population. Features (Particularities) RS7 Intelli IT (HEM-6232T-E) is an automatic wrist type blood pressure monitor that has the unique selling points as noted: •... -
Page 5: How To Use The Device
Review the Instruction Manual for the full details on the application of this product. Technical data Product category Electronic Sphygmomanometers Product Description Automatic Wrist Blood Pressure Monitor Model (code) RS7 Intelli IT (HEM-6232T-E) Display LCD Digital Display Cuff pressure range 0 to 299 mmHg Blood pressure SYS: 60 to 260 mmHg... - Page 6 Weight Approximately 91 g not including batteries Approximately 91 mm (w) × 63 mm (h) × 13 mm* (l) Dimensions (not including the wrist cuff) Measurable wrist 13.5 to 21.5 cm circumference Memory Stores up to 100 readings per user Monitor, storage case, 2 “AAA”...
- Page 7 Materials used in main device and all accessories Parts Name Material Acrylonitrile butadiene styrene Front Housing Polymethyl methacrylate Ground Housing Acrylonitrile butadiene styrene Battery Cover Acrylonitrile butadiene styrene Acrylonitrile butadiene styrene Button Polymethyl methacrylate Glass Robber key Silicone Rubber Polyester Cloth Bag Polyvinyl chloride Air bag...
- Page 8 Title page of Instruction Manual PM-1771-E-01-03/2018...
- Page 9 Unfolded package design of main device and accessories Main device: Accessories: Packaging specifications from Explanation Diagram Approx. dimensions Package Amount Approx. weight W x D x H, mm Main Unit 91 x 13.4 x 63.4 mm Package 297g 90 x 90 x 115 mm Master Carton 3.2kg 472 x 198 x 144 mm...
-
Page 10: List Of Harmonized En Standards
List of Harmonized EN Standards The device is classified as a medical device, Class IIa (MDD Annex IX Rule 10) Applicable Medical Device Directive (93/42/EEC) standards: EN ISO 15223-1:2016 EN 1041:2008 EN 1060ꞏ1:1995+A2:2009 EN 1060ꞏ3:1997+A2:2009 EN 60601-1:2006+A1:2013 EN 60601-1-2:2015 EN 60601-1-6:2010+A1:2015 EN 60601-1-11:2015 EN 80601-2-30:2010+A1:2015 EN 62304:2006+A1:2015... - Page 11 • DO NOT use this monitor on infants, toddlers, children or persons who cannot express themselves. • DO NOT adjust medication based on readings from this blood pressure monitor. Take medication as prescribed by your physician. ONLY a physician is qualified to diagnose and treat high blood pressure.
- Page 12 • Ensure that this monitor has acclimated to room temperature before taking a measurement. Taking a measurement after an extreme temperature change could lead to an inaccurate reading. OMRON recommends waiting for approximately 2 hours for the monitor to warm up or cool down when the monitor is used in an environment within the temperature specified as operating conditions after it is stored either at the maximum or at the minimum storage temperature.
-
Page 13: Transportation And Storage Conditions
• ONLY use 2 “AAA” alkaline batteries with this monitor. DO NOT use other types of batteries. DO NOT use new and used batteries together. DO NOT use different brands of batteries together. • Remove batteries if this monitor will not be used for a long period of time. •... - Page 14 Do not disassemble or attempt to repair the device or components. Consult your OMRON retail outlet or distributor. Servicing It is generally recommended to have the device inspected every 2 years to ensure correct functioning and accuracy. Please consult your OMRON retail outlet or distributor. PM-1771-E-01-03/2018...














Need help?
Do you have a question about the RS7 Intelli IT and is the answer not in the manual?
Questions and answers