Table of Contents
Advertisement
Quick Links
Advertisement
Table of Contents

Subscribe to Our Youtube Channel
Summary of Contents for CELIMED CL3
- Page 1 REV.: CELIMED CL3 (TMB-2084-N)- Návod k obsluze (model TMB-2084-N)
- Page 3 Obsah ÚVOD ..............2 .
- Page 8 POPIS...
- Page 9 ½...
- Page 10 kabel slabá baterie...
- Page 24 Systolický Diastolický...
- Page 26 NÁPRAVA...
- Page 29 IEC 61000-3-2 IEC 61000-3-3...
- Page 30 IEC 60601-1-2 IEC 61000-4-4 IEC61000-4-5 IEC 61000-4-11 IEC 61000-4-8 IEC61000-4-6 IEC61000-4-3...
- Page 31 1720 1845 2450 5240 5500 5785...
- Page 32 District, 528437 Zhongshan, Guangdong, Čína Zástupce pro EU: Medical Device Safety Service GmbH Schiffgraben 41, 30175 Hannover, Německo Dovozce pro ČR, Celimed s.r.o. autorizovaný servis: Sociální péče 3487/5a, 400 11 Ústí nad Labem, Česká republika Pozáruční servis – tel.: +420 475 208 180 e-mail: info@celimed.cz www.celimed.cz...
- Page 33 REV.: CELIMED CL3 (TMB-2084-N)- (model TMB-2084-N)
- Page 35 Obsah ÚVOD ..............34 .
- Page 40 OPIS...
- Page 42 kábel slabá batéria...
- Page 56 Systolický Diastolický tepna stiahnutie...
- Page 58 PREJAV NÁPRAVA...
- Page 61 IEC 61000-3-2 IEC 61000-3-3...
- Page 62 IEC 60601-1-2 IEC 61000-4-4 IEC61000-4-5 IEC 61000-4-11 IEC 61000-4-8 IEC61000-4-6 IEC61000-4-3...
- Page 63 KRYTOM PRÍSTROJA 1720 1845 1970 2450 5240 5500 5785...
- Page 64 District, 528437 Zhongshan, Guangdong, Čína Zástupca pre EÚ: Medical Device Safety Service GmbH Schiffgraben 41, 30175 Hannover, Nemecko Dovozca pre SR, Celimed s.r.o. autorizovaný servis: Pri majeri 22 831 06 Bratislava, Slovenská Republika Servis: 02 4468 1249 e-mail: info@celimed.sk www.celimed.sk...
-
Page 65: Felhasználói Kézikönyv
REV.: CELIMED CL3 (TMB-2084-N)- Felhasználói kézikönyv (model TMB-2084-N) Felkaros típus... - Page 67 KATALÓGUS...
- Page 68 BEVEZETÉS...
- Page 70 VIGYÁZA...
- Page 71 VIGYÁZA...
- Page 73 MANDZSETTA LÉGVEZETÉK ELEMTARTÓ REKESZ Lista alkatrész) 4. AC adapter BLJ06 L050100U-V 5. USB kábel 3. 4× AAA ELEM 6. Tárolótasak 7. Felhasználói kézikönyv...
- Page 74 AC adapter USB kábel VIGYÁZAT VIGYÁZAT...
- Page 79 MÉRÉS...
- Page 80 A jelenlegi felhasználó villog. A mért eredmény „ “ villog...
- Page 82 ADATKEZELÉS...
- Page 83 A jelenlegi memóriabejegyzés az 1. csoport. 06:00.
- Page 84 A „DEL YES” villog A „DEL no” villog A törlés megtörtént...
- Page 85 „DEL ALL” villog A „DEL no” villog A törlés megtörtént Nincs adat...
- Page 86 SZÁMÁRA Amikor beszél vagy mozgatja Nagyon hideg környezetben Amikor vizelnie kell...
- Page 87 Tartsa száraz helyen és óvja...
- Page 88 Szisztolé Diasztol a vér bejutása a vér kiürülése véna artéria megfes ellazul 3. fokozatú hipertónia (súlyos) 2. fokozatú hipertónia (mérsékelt) 1. fokozatú hipertónia (enyhe) Alcsoport: határeset Magas normális vérnyomás Normál vérnyomás Optimális vérnyomás Szisztolés vérnyomás (mmHg)
- Page 90 HIBAELHÁRÍTÁS...
- Page 92 Adapter Típus: BLJ06L050100U-V Bemenet: 100–240 V, 50–60 Hz, 0,2A max 1000 mA , 5,0 W...
- Page 93 EMC ÚTMUTATÓ Útmutató és gyártói nyilatkozat – elektromágneses kisugárzás Kibocs t si vizsg lat Megfelel s CISPR 11 [ B ] oszt ly CISPR 11 A oszt ly IEC 61000-3-2 Fesz lts IEC 61000-3-3...
- Page 94 2. táblázat Útmutató és gyártói nyilatkozat -– IEC 60601-1-2 Immunitásvizsgálat Megfelelési szint Vizsgálati szint ±8 kV érintkezés ±8 kV érintkezés kisülés (ESD) ±2 kV, IEC 61000-4-2 ±2 kV a tápvezetékek esetében ±2 kV a tápvezetékek esetében Gyors elektromos tranziens/kitörés ±1 kV jel bemenet/kimenet Nem alkalmazható...
- Page 95 3. táblázat Útmutató és gyártói nyilatkozat – Kibocsátott RF Vizsgálati Sáv (MHz) Szolgáltatás Maximális Távolság IEC 60601-1-2 teljesítmé Vizsgálati szint ési szint IEC61000-4-3 (V/m) (Vizsgálati (V/m) a (MHz) ny (W) 380-390 TETRA 400 Impulzus 18 Hz E a vezeték FM ± 5k Hz 430-470 GMRS 460, nélküli...
- Page 96 District, 528437 Zhongshan, Guangdong, Kína EU-kepviselet: Medical Device Safety Service GmbH Schiffgraben 41, 30175 Hannover, Németország Forgalmazo / HU Celimed s.r.o. importer Sociální péče 3487/5a, 400 11 Ústí nad Labem, Cseh Köztársaság Garancia utáni szerviz – tel.: +420 475 208 180 e-mail: info@celimed.cz www.celimed.cz...
-
Page 97: Blood Pressure Monitor
REV.: CELIMED CL3 (TMB-2084-N)- USER MANUAL Blood Pressure Monitor (model TMB-2084-N) Arm Type... - Page 99 CATALOGUE Table of Contents...
-
Page 100: General Description
INTRODUCTION General Description Indications for Use Contraindications Measurement Principle... -
Page 101: Safety Information
Safety Information EC REP... - Page 102 CAUTION * This device is intended for indoor, home use. * This device is not intended for public use. * This device is portable, but it is not intended for use during patient transport. * This device is not suitable for continuous monitoring during medical emergencies or operations. * This device is intended for no-invasive measuring and monitoring of arterial blood pressure.
- Page 103 CAUTION * Warning: This device is not AP/APG equipment. Do not use the device where flammable anesthetic are present, or in environments mixture with air of with oxygen or nitrous oxide. * The device contains sensitive electronic components.To avoid measurement errors, avoid taking blood pressure measurements near a strong electromagnetic field radiated interference signal or electrical fast transient/burst signal.
-
Page 104: Lcd Display Signal
LCD display signal SYMBOL DESCRIPTION EXPLANATION... -
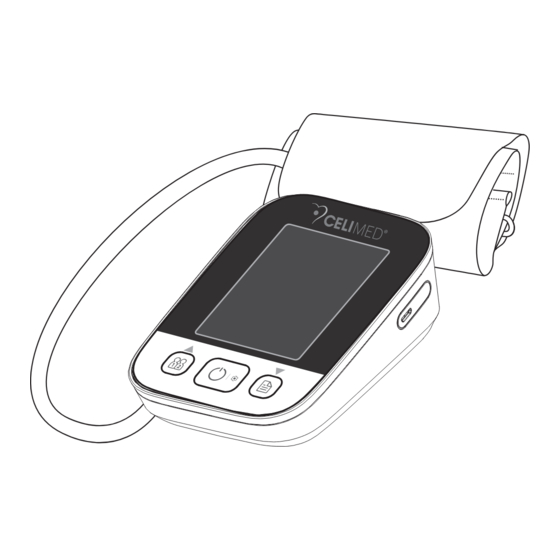
Page 105: Monitor Components
Monitor Components CUFF AIR HOSE USB CHARGING USER BUTTON BUTTON BUTTON List BLJ06L050100U-V 3. 4× 5. USB cable 7. User manual... -
Page 106: Installing And Replacing The Batteries
The Choice of Power Supply CAUTION Installing and Replacing the Batteries CAUTION... -
Page 107: Setting The Date And Time
Setting the Date and Time It is important to set the clock before using your blood pressure is stored in the memory. (year: 2021—2050, Time format: 12H/24H) -
Page 110: Select The User Id
Select the User ID Note:... -
Page 111: Tie The Cuff
MEASUREMENT Tie the cuff... -
Page 112: Start The Measurement
Start the Measurement... -
Page 114: Data Management
DATA MANAGEMENT Recall the Records... - Page 115 DATA MANAGEMENT CAUTION...
-
Page 116: Delete The Records
Delete the Records will display “dEL do nE” the unit... -
Page 118: Information For User
INFORMATION FOR USER Tips for Measurement... -
Page 119: Maintenance
Maintenance... -
Page 120: What Is The Standard Blood Pressure Classification
What are systolic pressure and diastolic pressure? Systolic Diastolic blood entering blood discharging vein artery press relax What is the standard blood pressure classification? CAUTION Irregular Heartbeat Detector CAUTION... -
Page 121: Why Does My Blood Pressure Fluctuate Throughout The Day
Why does my blood pressure fluctuate throughout the day? Why do I get different blood pressure at home compared to the hospital? Is the result the same if measuring on the right arm? -
Page 122: Troubleshooting
TROUBLE SHOOTING If any abnormality arises during use, please check the following points: PROBLEM SYMPTOM CHECK THIS REMEDY No power High power batteries Error message Warning message... - Page 123 Power supply Display mode Measurement mode Measurement range Accuracy Normal working condition Storage & transportation condition Measurement perimeter of the upper arm Weight External dimensions Attachment Mode of operation Degree of protection Protection against ingress of water Device Classification Software Version...
-
Page 124: Athorized Component
Athorized Component Please use the TRANSTEK authorized adapter. Adapter Type: BLJ06L050100U-V Input: 100-240V, 50-60Hz, 0.2A max 1000 mA Output: Limited Warranty The manufacturer provides a 2-year warranty on this product (main unit) from Contact Information the date of purchase. -
Page 125: Emc Guidance
EMC GUIDANCE EMC Guidance The ME EQUIPMENT or ME SYSTEM is suitable for home healthcare environments. Warning: Don’t be near the active HF surgical equipment and the RF shielded room of an ME system for magnetic resonance imaging, where the intensity of EM disturbances is high. Warning: Use of this equipment adjacent to or stacked with other equipment should be avoided because it could result in improper operation. - Page 126 Table 2 Guidance and manufacturer’s declaration – electromagnetic Immunity IEC 60601-1-2 Immunity Test Compliance level Test level Electrostatic ±8 kV contact ±8 kV contact discharge (ESD) ±2 kV, ±4 kV, ±8 kV, ±15 kV air ±2 kV, ±4 kV, ±8 kV, ±15 kV air IEC 61000-4-2 ±2 kV for power supply lines ±2 kV for power supply lines...
- Page 127 Table 3 Guidance and manufacturer’s declaration - electromagnetic Immunity Compliance Radiated RF Test Band Service Modulation Distance Maximum 60601-1-2 level IEC61000-4-3 Frequency (MHz) Power Test Level (V/m) (MHz) (Test (V/m) specifications 380-390 Pulse TETRA modulation ENCLOSURE 18 Hz PORT IMMUNITY to 430-470 GMRS 460, FM ±...
- Page 128 China Representative for Medical Device Safety Service GmbH the EU: Schiffgraben 41, 30175 Hannover, Germany Importer for the Celimed s.r.o. CR, authorized Sociální péče 3487/5a, service: 400 11 Ústí nad Labem, Czech Republic Post-warranty service – tel.: +420 475 208 180 e-mail: info@celimed.cz www.celimed.cz...
Need help?
Do you have a question about the CL3 and is the answer not in the manual?
Questions and answers