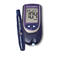
Guarantee - Accu-Chek Performa Owner's Booklet
Hide thumbs
Also See for Performa:
- Quick start manual ,
- Owner's booklet (228 pages) ,
- User manual (180 pages)
Table of Contents
Advertisement
Catalogue number
For in vitro diagnostic use. Do not ingest!
This product fulfills the requirements of Directive 98/79/EC on in vitro
diagnostic medical devices.
Listed by Underwriter's Laboratories, Inc.
and CAN/CSA C22.2 No.1010-1.
3V type 2032
Guarantee
The statutory guarantee provisions governing the sale of consumer goods in the country of
purchase shall prevail.
©
in accordance with UL 61010A-1
69
Hide quick links:
Advertisement
Table of Contents
Troubleshooting